18.7: Latent Heat of Fusion
- Page ID
- 8678
The most straightforward method for measuring the specific latent heat L of ice is to drop a lump of
Ice of mass m and specific latent heat L at its melting point T0 into a
Calorimeter of mass MC and specific heat capacity CC and initial (warm) temperature T2,
which contains
a mass MW of Water of specific heat capacity CW at the same warm temperature T2.
After the ice has melted everything comes to a final (cool) temperature T1. Then
\[m\left[L+C_{\mathrm{W}}\left(T_{2}-T_{0}\right)\right]=\left(M_{\mathrm{C}} C_{\mathrm{C}}+M_{\mathrm{w}} C_{\mathrm{w}}\right)\left(T_{2}-T_{1}\right).\]
If the temperatures in this equation are supposed to be in degrees Celsius, so that T0 = 0, and if masses are in grams and heat in calories, so that C2 = 1, this equation becomes
\[m\left(L+T_{2}\right)=\left(M_{C} C_{C}+M_{W}\right)\left(T_{2}-T_{1}\right).\]
For good results, heat losses must be minimized and allowed for, and precautions must be taken to minimize and allow for any water initially clinging to the lump of ice.
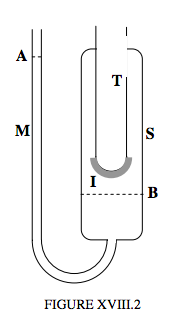
Quite precise measurements of the latent heat of fusion of ice can be made with the Bunsen Ice Calorimeter, an apparatus that can also be used to measure specific heat capacities of other substances. My limited artistic skills with the computer do not allow me to draw all the minute details of the practical construction of a Bunsen ice calorimeter that makes it a precision instrument, but may, perhaps, suffice to show the general principles, in figure XVIII.2. A test-tube T, is fitted with an outer glass sleeve S, the lower end of which leads to a manometer M. The portion of the sleeve above the level B is filled with air-free pure water at its freezing point. The manometer from level A to level B is filled with mercury. The entire apparatus is generally enclosed in a large ice-box, so that the entire apparatus is at 0°C. Some ice is formed outside the bottom of the test-tube, at I. In order to measure the specific latent heat of fusion of ice, a measured quantity of hot water is poured into test-tube. This water, in cooling down to 0°C, gives up a known amount of heat to the ice, some of which melts. So – how do you know how much ice has melted? Water ice contracts on melting into liquid. Consequently the level B moves up and the level A (which can be in a quite narrow capillary tube) goes down, so the reduction in volume (and hence the mass of ice melted) can be determined quite accurately. Thus the latent heat of fusion of ice can be determined. Once the equipment has been calibrated (i.e. when we know how much movement of level A corresponds to how much transfer of heat), the calorimeter can be used to measure specific heat capacities of other substances, simply by dropping a known mass of the substance at a known temperature into the test-tube, and measuring the movement of the level A. It will be understood, I think, that, in using the apparatus to measure the specific latent heat of ice, it is necessary to know the densities of ice and of water precisely. To use it for measuring the specific heat capacities of other substances, it is not necessary to know this, or even to know the specific latent heat of fusion of ice. You do have to know the specific heat capacity of water – which is not much of a burden, especially if you are content to express heat in calories!