11.8: Cohesion and Adhesion in Liquids - Surface Tension and Capillary Action
- Page ID
- 1568
Learning Objectives
By the end of this section, you will be able to:
- Understand cohesive and adhesive forces.
- Define surface tension.
- Understand capillary action.
Children blow soap bubbles and play in the spray of a sprinkler on a hot summer day (Figure \(\PageIndex{1}\)). An underwater spider keeps his air supply in a shiny bubble he carries wrapped around him. A technician draws blood into a small-diameter tube just by touching it to a drop on a pricked finger. A premature infant struggles to inflate her lungs. What is the common thread? All these activities are dominated by the attractive forces between atoms and molecules in liquids—both within a liquid and between the liquid and its surroundings.

Attractive forces between molecules of the same type are called cohesive forces. Liquids can, for example, be held in open containers because cohesive forces hold the molecules together. Attractive forces between molecules of different types are called adhesive forces. Such forces cause liquid drops to cling to window panes, for example. In this section we examine effects directly attributable to cohesive and adhesive forces in liquids.
Definition: Cohesive Forces
Attractive forces between molecules of the same type are called cohesive forces.
Definition: Adhesive Forces
Attractive forces between molecules of different types are called adhesive forces.
Surface Tension
Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. This general effect is called surface tension. Molecules on the surface are pulled inward by cohesive forces, reducing the surface area. Molecules inside the liquid experience zero net force, since they have neighbors on all sides.
Definition: Surface Tension
Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. This general effect is called surface tension.
Surface Tension
Forces between atoms and molecules underlie the macroscopic effect called surface tension. These attractive forces pull the molecules closer together and tend to minimize the surface area. This is another example of a submicroscopic explanation for a macroscopic phenomenon.
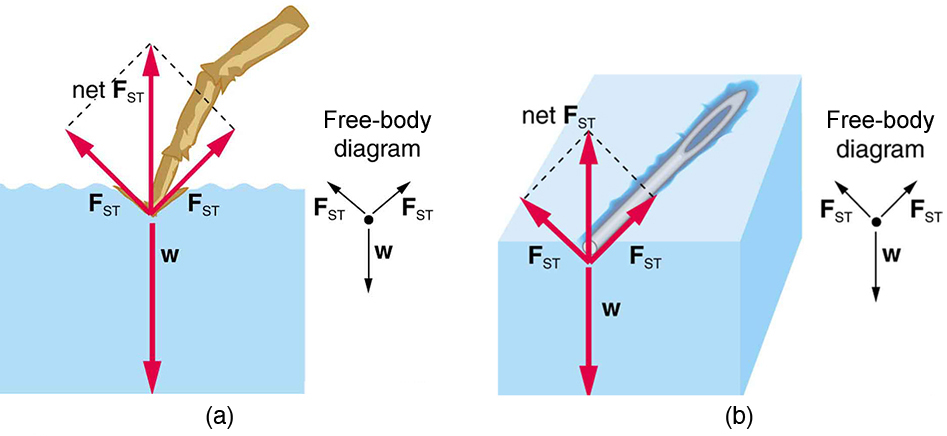
The model of a liquid surface acting like a stretched elastic sheet can effectively explain surface tension effects. For example, some insects can walk on water (as opposed to floating in it) as we would walk on a trampoline—they dent the surface as shown in Figure \(\PageIndex{2a}\). Figure \(\PageIndex{2b}\) shows another example, where a needle rests on a water surface. The iron needle cannot, and does not, float, because its density is greater than that of water. Rather, its weight is supported by forces in the stretched surface that try to make the surface smaller or flatter. If the needle were placed point down on the surface, its weight acting on a smaller area would break the surface, and it would sink.
Surface tension is proportional to the strength of the cohesive force, which varies with the type of liquid. Surface tension \(\overline{\gamma}\) is defined to be the force F per unit length \(L\) exerted by a stretched liquid membrane:
\[\gamma = \dfrac{F}{L}.\]
Table \(\PageIndex{1}\) lists values of \(\overline{\gamma}\) for some liquids.
| Liquid | Surface tension γ(N/m) |
|---|---|
| Water at \(0^oC\) | 0.0756 |
| Water at \(20^oC\) | 0.0728 |
| Water at \(100^OC\) | 0.0589 |
| Soapy water (typical) | 0.0370 |
| Ethyl alcohol | 0.0223 |
| Glycerin | 0.0631 |
| Mercury | 0.465 |
| Olive Oil | 0.032 |
| Tissue fluids (typical) | 0.050 |
| Blood, whole at \(37^oC\) | 0.058 |
| Blood plasma at \(37^oC\) | 0.073 |
| Gold at \(1070^oC\) | 1.000 |
| Oxygen at \(-193^oC\) | 0.0157 |
| Helium at \(-269^oC\) | 0.00012 |
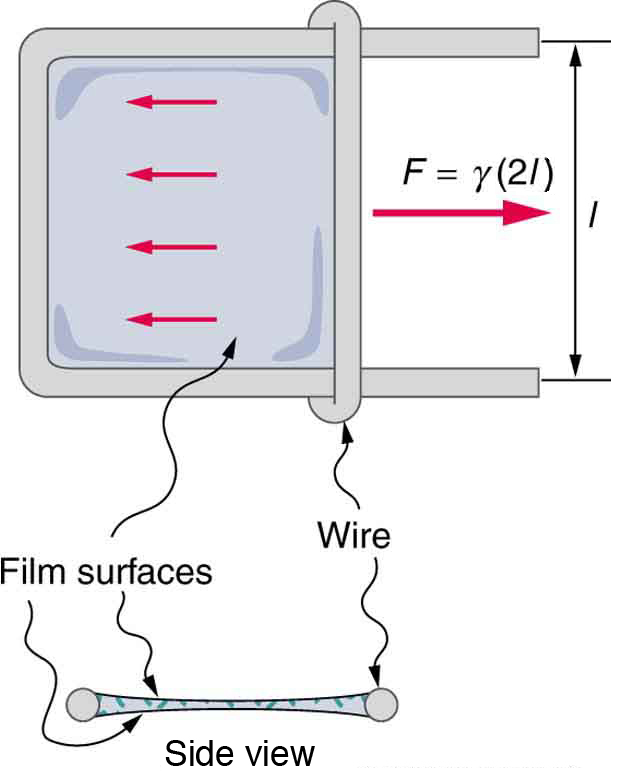
For the insect of Figure Figure \(\PageIndex{1a}\), its weight \(w\) is supported by the upward components of the surface tension force: \(w = \gamma L \sin \theta\), where \(L\) is the circumference of the insect’s foot in contact with the water. Figure \(\PageIndex{3}\) shows one way to measure surface tension. The liquid film exerts a force on the movable wire in an attempt to reduce its surface area. The magnitude of this force depends on the surface tension of the liquid and can be measured accurately.
Surface tension is the reason why liquids form bubbles and droplets. The inward surface tension force causes bubbles to be approximately spherical and raises the pressure of the gas trapped inside relative to atmospheric pressure outside. It can be shown that the gauge pressure \(P \) inside a spherical bubble is given by
\[P = \dfrac{4\gamma}{r},\]
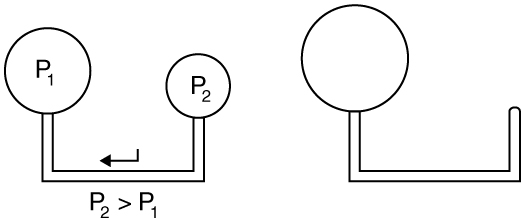
where \(r\) is the radius of the bubble. Thus the pressure inside a bubble is greatest when the bubble is the smallest. Another bit of evidence for this is illustrated in Figure \(\PageIndex{4}\). When air is allowed to flow between two balloons of unequal size, the smaller balloon tends to collapse, filling the larger balloon.
Example \(\PageIndex{1}\): Surface Tension: Pressure Inside a Bubble
Calculate the gauge pressure inside a soap bubble \(2.00 \times 10^{-4} m\) in radius using the surface tension for soapy water in Table. Convert this pressure to mm Hg.
Strategy
The radius is given and the surface tension can be found in Table, and so \(P\) can be found directly from the equation \(P = \frac{4\gamma}{r}\).
Solution
Substituding \(r\) and \(\gamma\) into this equation \(P = \frac{4\gamma}{r}\), we obtaiin
\[P = \dfrac{4\gamma}{r} = \dfrac{4(0.037 \, N/m)}{2.00 \times 10^{-4}m} = 740 \, N/m^2 = 740 \, Pa.\]
We use a conversion factor to get this into units of mm Hg:
\[P = (740 \, N/m^2)\dfrac{1.00 \, mm \, Hg}{133 \, N/m^2} = 5.56 \, mm \, Hg.\]
Discussion
Note that if a hole were to be made in the bubble, the air would be forced out, the bubble would decrease in radius, and the gauge pressure would reduce to zero, and the absolute pressure inside would decrease to atmospheric pressure (760 mm Hg).
Our lungs contain hundreds of millions of mucus-lined sacs called alveoli, which are very similar in size, and about 0.1 mm in diameter. (See Figure.) You can exhale without muscle action by allowing surface tension to contract these sacs. Medical patients whose breathing is aided by a positive pressure respirator have air blown into the lungs, but are generally allowed to exhale on their own. Even if there is paralysis, surface tension in the alveoli will expel air from the lungs. Since pressure increases as the radii of the alveoli decrease, an occasional deep cleansing breath is needed to fully reinflate the alveoli. Respirators are programmed to do this and we find it natural, as do our companion dogs and cats, to take a cleansing breath before settling into a nap.
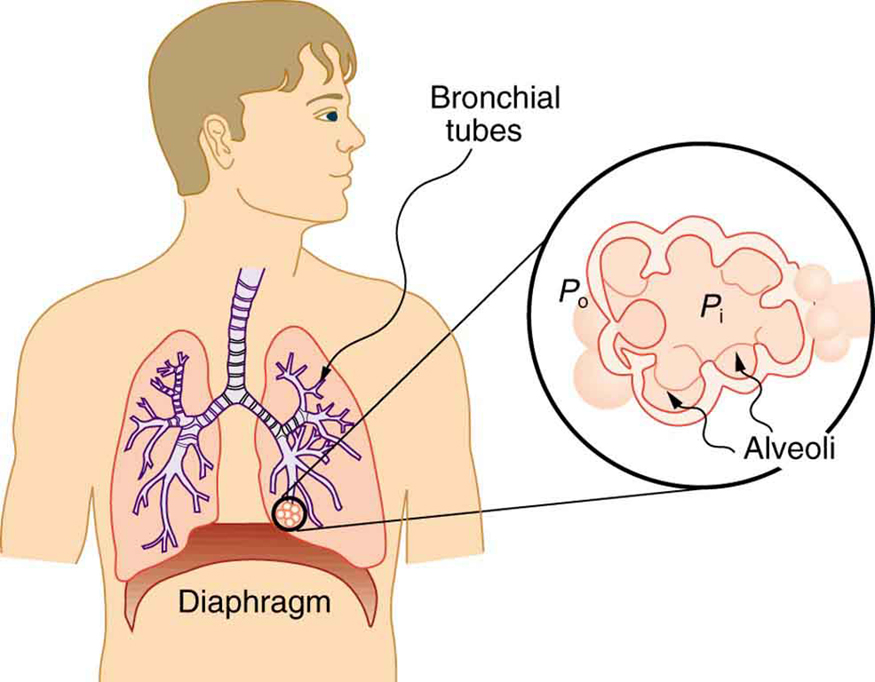
The tension in the walls of the alveoli results from the membrane tissue and a liquid on the walls of the alveoli containing a long lipoprotein that acts as a surfactant (a surface-tension reducing substance). The need for the surfactant results from the tendency of small alveoli to collapse and the air to fill into the larger alveoli making them even larger (as demonstrated in Figure). During inhalation, the lipoprotein molecules are pulled apart and the wall tension increases as the radius increases (increased surface tension). During exhalation, the molecules slide back together and the surface tension decreases, helping to prevent a collapse of the alveoli. The surfactant therefore serves to change the wall tension so that small alveoli don’t collapse and large alveoli are prevented from expanding too much. This tension change is a unique property of these surfactants, and is not shared by detergents (which simply lower surface tension). (See Figure.)
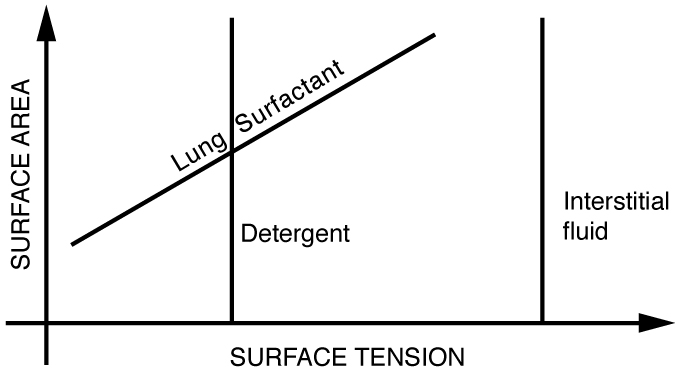
If water gets into the lungs, the surface tension is too great and you cannot inhale. This is a severe problem in resuscitating drowning victims. A similar problem occurs in newborn infants who are born without this surfactant—their lungs are very difficult to inflate. This condition is known as hyaline membrane disease and is a leading cause of death for infants, particularly in premature births. Some success has been achieved in treating hyaline membrane disease by spraying a surfactant into the infant’s breathing passages. Emphysema produces the opposite problem with alveoli. Alveolar walls of emphysema victims deteriorate, and the sacs combine to form larger sacs. Because pressure produced by surface tension decreases with increasing radius, these larger sacs produce smaller pressure, reducing the ability of emphysema victims to exhale. A common test for emphysema is to measure the pressure and volume of air that can be exhaled.
Making Connections: Take-Home Investigation
- Try floating a sewing needle on water. In order for this activity to work, the needle needs to be very clean as even the oil from your fingers can be sufficient to affect the surface properties of the needle.
- Place the bristles of a paint brush into water. Pull the brush out and notice that for a short while, the bristles will stick together. The surface tension of the water surrounding the bristles is sufficient to hold the bristles together. As the bristles dry out, the surface tension effect dissipates.
- Place a loop of thread on the surface of still water in such a way that all of the thread is in contact with the water. Note the shape of the loop. Now place a drop of detergent into the middle of the loop. What happens to the shape of the loop? Why?
- Sprinkle pepper onto the surface of water. Add a drop of detergent. What happens? Why?
- Float two matches parallel to each other and add a drop of detergent between them. What happens? Note: For each new experiment, the water needs to be replaced and the bowl washed to free it of any residual detergent.
Adhesion and Capillary Action
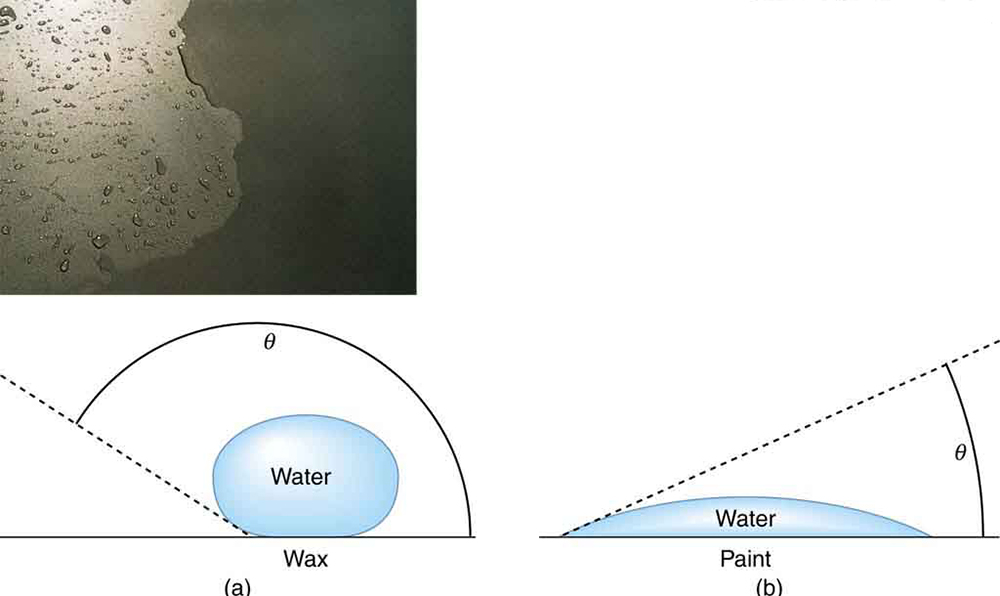
Why is it that water beads up on a waxed car but does not on bare paint? The answer is that the adhesive forces between water and wax are much smaller than those between water and paint. Competition between the forces of adhesion and cohesion are important in the macroscopic behavior of liquids. An important factor in studying the roles of these two forces is the angle \(\theta\) between the tangent to the liquid surface and the surface (Figure \(\PageIndex{7}\)). The contact angle \(\theta\) is directly related to the relative strength of the cohesive and adhesive forces. The larger the strength of the cohesive force relative to the adhesive force, the larger \(\theta\) is, and the more the liquid tends to form a droplet. The smaller \(\theta\) is, the smaller the relative strength, so that the adhesive force is able to flatten the drop. Table lists contact angles for several combinations of liquids and solids.
Definition: Contact Angle
The angle \(\theta\) between the tangent to the liquid surface and the surface is called the contact angle.
One important phenomenon related to the relative strength of cohesive and adhesive forces is capillary action—the tendency of a fluid to be raised or suppressed in a narrow tube, or capillary tube. This action causes blood to be drawn into a small-diameter tube when the tube touches a drop.
Capillary Action
The tendency of a fluid to be raised or suppressed in a narrow tube, or capillary tube, is called capillary action.
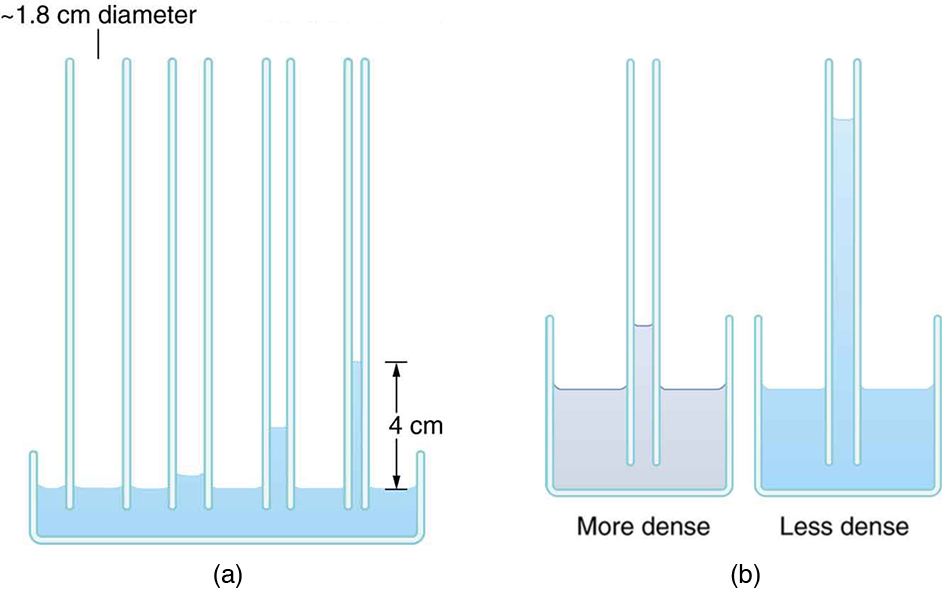
If a capillary tube is placed vertically into a liquid, as shown in Figure, capillary action will raise or suppress the liquid inside the tube depending on the combination of substances. The actual effect depends on the relative strength of the cohesive and adhesive forces and, thus, the contact angle \(\theta\) given in the table. If \(\theta\) is less that \(90^o\), then the fluid will be raised; if \(\theta\) is greater than \(90^o\), it will be suppressed. Mercury, for example, has a very large surface tension and a large contact angle with glass. When placed in a tube, the surface of a column of mercury curves downward, somewhat like a drop. The curved surface of a fluid in a tube is called a meniscus. The tendency of surface tension is always to reduce the surface area. Surface tension thus flattens the curved liquid surface in a capillary tube. This results in a downward force in mercury and an upward force in water, as seen in Figure.

| Interface | Contact angle \(\phi\) |
|---|---|
| Mercury-glass | \(140^o\) |
| Water-glass | \(0^o\) |
| Water-paraffin | \(107^o\) |
| Water-silver | \(90^o\) |
| Organic liquids (most)-glass | \(0^o\) |
| Ethyl alcohol-glass | \(0^o\) |
| Kerosene-glass | \(26^o\) |
Capillary action can move liquids horizontally over very large distances, but the height to which it can raise or suppress a liquid in a tube is limited by its weight. It can be shown that this heigh \(h\) is given by
\[h = \dfrac{2\gamma \cos \theta}{\rho gr}.\]
If we look at the different factors in this expression, we might see how it makes good sense. The height is directly proportional to the surface tension \(\gamma\), which is its direct cause. Furthermore, the height is inversely proportional to tube radius—the smaller the radius , the higher the fluid can be raised, since a smaller tube holds less mass. The height is also inversely proportional to fluid density , since a larger density means a greater mass in the same volume. (See Figure.)
Example \(\PageIndex{2}\): Calculating Radius of a Capillary Tube: Capillary Action: Tree Sap
Can capillary action be solely responsible for sap rising in trees? To answer this question, calculate the radius of a capillary tube that would raise sap 100 m to the top of a giant redwood, assuming that sap’s density is \(1050 \, kg/m^3\), its contact angle is zero, and its surface tension is the same as that of water at \(20.0^oC\).
Strategy
The height to which a liquid will rise as a result of capillary action is given by \(h = \frac{2\gamma \cos \theta}{\rho gr}\), and every quantity is known exceptfor \(r\).
Solution
Solving for \(r\) and substituting known values produces
\[r = \dfrac{2\gamma \cos \theta}{\rho gh} = \dfrac{2(0.0728 \, N/m)cos(0^o)}{(1050 \, kg/m^3)(9.80 \, m/s^2)(100 \, m)}\]
\[= 1.41 \times 10^{-7} \, m.\]
Discussion
This result is unreasonable. Sap in trees moves through the xylem, which forms tubes with radii as small as \(2.5 \times 10^{-7} \, m.\) This value is about 180 times as large as the radius found necessary here to raise sap \(100 m\). This means that capillary action alone cannot be solely responsible for sap getting to the tops of trees.
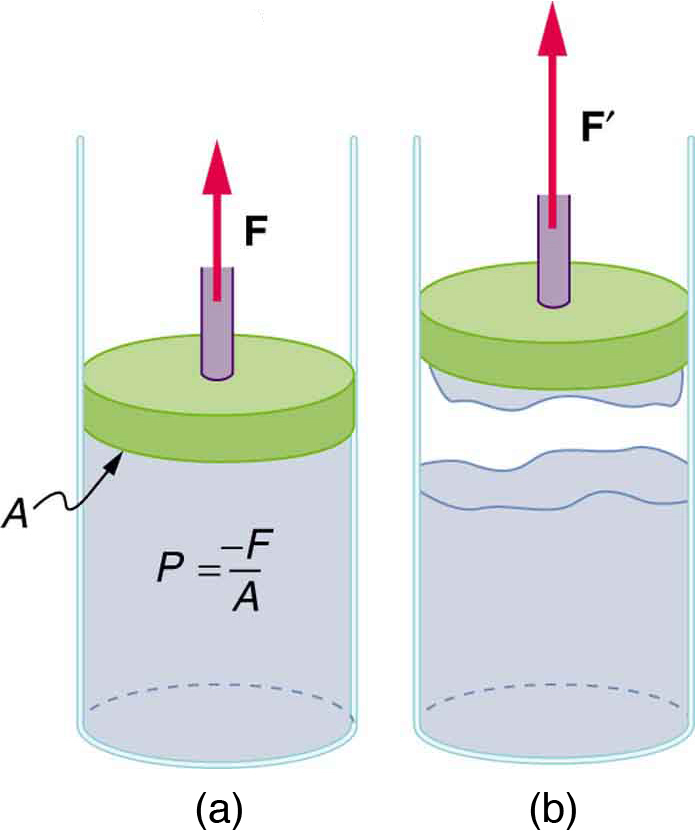
How does sap get to the tops of tall trees? (Recall that a column of water can only rise to a height of 10 m when there is a vacuum at the top—see [link].) The question has not been completely resolved, but it appears that it is pulled up like a chain held together by cohesive forces. As each molecule of sap enters a leaf and evaporates (a process called transpiration), the entire chain is pulled up a notch. So a negative pressure created by water evaporation must be present to pull the sap up through the xylem vessels. In most situations, fluids can push but can exert only negligible pull, because the cohesive forces seem to be too small to hold the molecules tightly together. But in this case, the cohesive force of water molecules provides a very strong pull. Figure shows one device for studying negative pressure. Some experiments have demonstrated that negative pressures sufficient to pull sap to the tops of the tallest trees can be achieved.
Summary
- Attractive forces between molecules of the same type are called cohesive forces.
- Attractive forces between molecules of different types are called adhesive forces.
- Cohesive forces between molecules cause the surface of a liquid to contract to the smallest possible surface area. This general effect is called surface tension.
- Capillary action is the tendency of a fluid to be raised or suppressed in a narrow tube, or capillary tube which is due to the relative strength of cohesive and adhesive forces.
Glossary
- adhesive forces
- the attractive forces between molecules of different types
- capillary action
- the tendency of a fluid to be raised or lowered in a narrow tube
- cohesive forces
- the attractive forces between molecules of the same type
- contact angle
- the angle \(θ\) between the tangent to the liquid surface and the surface
- surface tension
- the cohesive forces between molecules which cause the surface of a liquid to contract to the smallest possible surface area


