5.1: Bragg Diffraction
( \newcommand{\kernel}{\mathrm{null}\,}\)
You should immediately ask, “How was the wave-like nature of matter experimentally verified?” If matter has a wave-like nature, it should exhibit interference in a manner completely analogous to the interference of light. Thus, when passing through a regular array of slits, or reflecting from a regular array of atoms, an interference pattern should form. In 1927 Clinton Davisson and David Germer tested this hypothesis by directing a beam of electrons at a crystal of nickel.
Incoming waves reflecting from the first crystal plane will interfere with waves reflecting from the second (and subsequent) crystal planes forming an interference pattern. This interference, termed Bragg diffraction, had been initially investigated using x-rays.
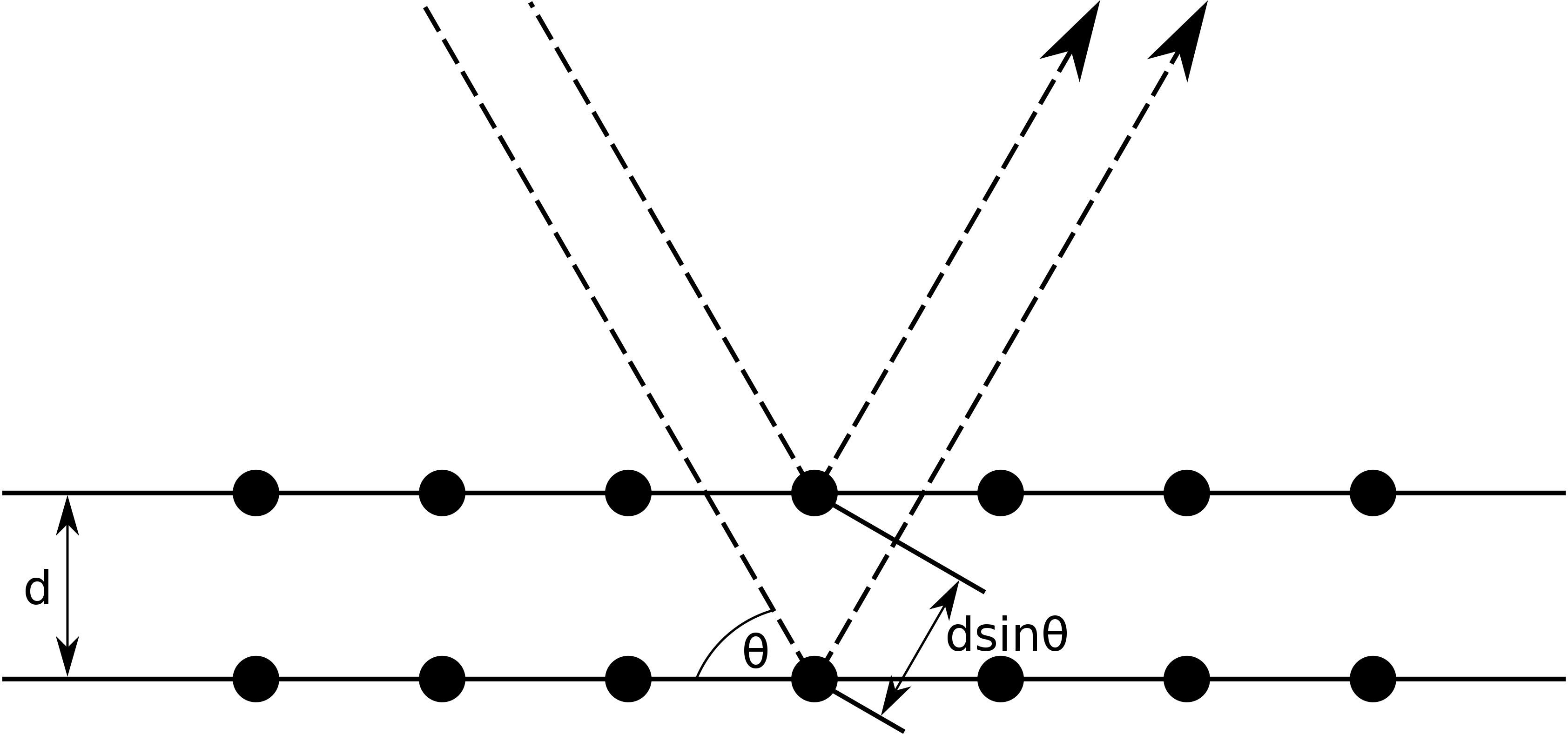
For constructive interference, the path length difference between the two reflected beams must differ by an integer multiple of a complete wavelength. From the diagram above, the wave reflecting from the second crystal plane travels an additional distance of \(2d \sin q\(. Thus, the relation for constructive interference is:
2dsinθ−nλ
where
- d is the distance between adjacent crystal planes, termed the lattice spacing,
- θ is the angle, measured from the crystal face, at which constructive interference occurs,
- and l is the wavelength of the disturbance.
A beam of electrons is accelerated through a potential difference of 54 V and is incident on a nickel crystal. The primary interference maximum is detected at 13.7o from the crystal face. What is the lattice spacing of the crystal?
If a beam of electrons is accelerated through a potential difference of 54 V, it gains a kinetic energy of 54 eV. This results in a momentum of
lE2total=(pc)2+(mc2)2
pc=√E2total−(mc2)2
pc=√54+511000)2−(511000)2
pc−7.43 keV
and, by DeBroglie’s relation, a wavelength of
λ=hcpc
λ=1240 eVnm7430 eV
λ=0.167 nm
Inserting this result into the Bragg relation results in
2dsinθ=nλ
2dsin13.7=(1)(0.167 nm)
d=0.352\texTnm
This value agrees with the known lattice spacing of nickel.
The presence of distinct interference maxima validates the idea that matter has a wave-like nature, and the agreement in lattice spacing illustrates that DeBroglie’s relationship between the momentum and wavelength of matter is correct. For their experimental validation of DeBroglie’s relation, Davisson (but not poor Mr. Germer) was awarded the Nobel Prize in 1937.


