6.7: Bose-Einstein Statistics
( \newcommand{\kernel}{\mathrm{null}\,}\)
Perhaps you thought the Fermi-Dirac results were strange: Non-interacting particles forming a collection as hard as steel. . . room temperature being effectively zero. Wait until you see the Bose-Einstein results.
6.7.1 Theory
For independent bosons, whether free or subject to an external potential, the mean occupation number function is
b(E)=1eβ(E−μ)−1.
To begin to understand this function, note that
when: we have: E<μb(E) negative E=μb(E)=∞E>μb(E) positive
Thus this function has the general character sketched below:
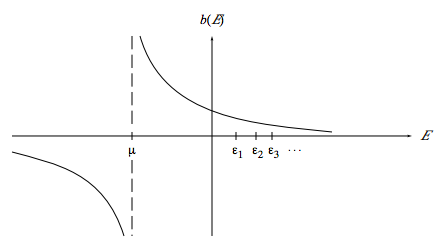
Although we have written b(E) as a function of the continuous variable E, we will in fact have occasion to evaluate it only at the energy eigenvalues ϵ1,ϵ2,ϵ3,...,ϵr,..., eigenvalues which of course differ for different external potentials. It seems bizarre that b(E) can be negative, and indeed this is only a mathematical artifact: Recall that in our derivation of the Bose function we needed to assume that µ < ε1 in order to insure convergence (see equation 6.35). Evaluating b(E) at any eigenvalue r will always result in a positive mean occupation number.
The character of the Bose function is dominated by the singularity at E = µ, so in trying to understand the function and its physical implications one must first locate the chemical potential. This section will provide a tour of Bose-Einstein behavior with decreasing temperature, throughout which the chemical potential shifts to the right. (When we investigated Fermi-Dirac behavior, we started at T = 0 and toured with increasing temperature, so the chemical potential shifted left.) This rightward shift presents a potential problem, because as the temperature decreases µ might shift right all the way to ϵ1, and we know that µ < 1. We will just have to go ahead and take the tour, being wary because we know that a µ = 1 road block might pop up right in front of us as we view the countryside. With any luck µ will not yet have reached ϵ1 when our tour halts at T = 0.
For the case of free and independent bosons (subject to periodic boundary conditions), the ground level energy is ϵ1 = 0. The natural first step is to find µ(T, V, N) by demanding that
N=∫∞0G(E)b(E)dE.
Natural though this may be, caution is in order. Remember that the integral above is an approximation to the sum over discrete energy levels
N=∑rb(ϵr).
It is legitimate to replace the sum with the integral when the value of b(E) changes little from one energy level to the next. We saw in section 6.5 that in the thermodynamic limit, the free-particle level spacings approach zero, so for the most part this approximation is excellent. . . even exact in the thermodynamic limit. But there is one exception: At ϵ = µ, the Bose function b(E) is infinite, so b(µ) = ∞ is very different from b(µ + δ), no matter how small the positive number δ is. Usually we can ignore this caution, because µ < 1 = 0. But if µ approaches 0 then we expect the approximation (6.71) to fail.
In summary, the integral (6.71) is a good approximation for the sum (6.72) as long as the integrand varies slowly. Now for any value of E greater than µ, you can make b(E + δ) very close to b(E) simply by choosing δ > 0 small enough. This is what happens in the thermodynamic limit. But for E = µ, there is always a large difference between b(E) = ∞ and b(E + δ), which is finite. Thus the integral approximation will be a good one as long as we avoid E = µ.
In situations where the sum can be legitimately replaced with the integral, we have
N=∫∞0G(E)b(E)dE
=V[√2m32π2ℏ3]∫∞0√E1eβ(E−μ)−1dE.
Use of the obvious substitution x=βE gives
N=V[√2m32π2ℏ3](kBT)3/2∫∞0x1/2exe−βμ−1dx,
and remembering the definition (5.4) of thermal de Broglie wavelength results in a more convenient expression
N=2√πVλ3(T)∫∞0x1/2exe−βμ−1dx.
Note that the definite integral above is not “just a number”. . . it is a function of the product βµ. There is no closed-form expression for the integral, although it is readily evaluated numerically and can be found in tabulations of functions. However it is easy to find an analytic upper bound: Because µ < 0, we have e−βµ > 1 whence
∫∞0x1/2exe−βμ−1dx<∫∞0x1/2ex−1dx.
The expression on the right is just a number, and a little bit of work (see problem 6.26) shows that it is the number
ζ(32)√π2,
where the Riemann zeta function is defined by
ζ(s)=∞∑n=11ns and ζ(32)=2.612375348…
So, how does this upper bound help us? It shows that
N=2√πVλ3(T)∫∞0x1/2exe−βμ−1dx<Vλ3(T)ζ(32),
but what is the significance of this result? Remember that the upper bound is just the value of the function when µ = 0, which is exactly where we expect a road block due to the breakdown of the integral approximation (6.71). Our hopes that we could avoid the issue have been dashed. The breakdown occurs at the temperature T0 that satisfies
N=Vλ3(T0)ζ(32) or λ3(T0)=ζ(32)/ρ
or
T0(ρ)=h22πmkBζ2/3(32)ρ2/3.
For temperatures above T0, the chemical potential shifts right as the temperature falls, and the integral approximation (6.71) is legitimate (indeed, exact in the thermodynamic limit). But as the temperature decreases below T0, the chemical potential sticks at µ = 0 and the integral approximation (6.71) must be modified.
What is the proper modification? The function b(E) is slowly varying for all values of E except E = µ, so the integral approximation is legitimate for all the energy levels except the ground level at 1 = 0 = µ. Only for the ground level is some other result needed, so we just add in the ground level occupancy by hand:
N=⟨n1⟩+∫∞0G(E)b(E)dE.
Now, we have already seen that when µ = 0—the condition for validity of this equation—the integral can be evaluated exactly and we have
N=⟨n1⟩+Vλ3(T)ζ(32).
Note that ⟨n1⟩ here is not given by its traditional formula (6.45), because
1eβ(E−μ)−1=1e0−1=∞.
Instead, equation (6.84) is the formula for ⟨n1⟩ when T < T0. The mean ground level occupancy ⟨n1⟩ is an intensive quantity when T > T0 but an extensive quantity when T < T0.
In summary, the correct normalization equation breaks into two parts, namely
N={2√πVλ3(T)∫∞0x1/2exe−βμ−1dx for T>T0(ρ)⟨n1⟩+Vλ3(T)ζ(32) for T<T0(ρ)
We should expect that each part will behave quite differently, i.e. we expect a sudden change of behavior as the temperature drops through T0.
What does all this mean physically? A naive reading of equation (6.80) suggests an upper bound on the number of particles that can be placed into the volume V. This would be sensible if the particles were marbles with hard-core repulsions. But these are non-interacting particles! Surely we can add more particles just by throwing them into the container. Indeed we can do so, and the associated excess mean occupation number is due to the level that particles like best at low temperatures, namely the ground level. The ground level thus has a much higher mean occupancy than the first excited level, and this rapid variation of mean occupancy with energy renders the approximation of sum by integral invalid. The inequality (6.80) does not limit the number of particles in the system: instead it shows the domain within which it is legitimate to approximate the sum (6.72) by the integral (6.71).
The abrupt transition at T0(ρ) is called Bose-Einstein condensation and the material at temperatures below T0(ρ) is called the Bose condensate. These terms are unfortunate: they conjure images of a gas condensing into a liquid, in which circumstance the atoms separate into two different classes: those in the liquid and those remaining in the vapor. This suggests that in Bose-Einstein condensation too there are two classes of particles: those in the ground level and those in the excited levels. This picture is totally false. It is incorrect to say “one particle is in the ground level, another is in the fourth excited level”. In truth the individual particles are not in individual levels at all: instead the whole system is in a state produced by multiplying together the individual level wavefunctions (“building blocks”) and then symmetrizing them. The literature of Bose-Einstein statistics is full of statements like “at temperatures below T0, any particle added to the system goes into the ground level.” Such statements are wrong. They should be replaced with statements like “at temperatures below T0, any increase in particle number occurs through an increase in ⟨n1⟩, the mean occupancy of the ground level.” Or alternatively, “at temperatures below T0, it is very likely that many of the building blocks from which the system wavefunction is built are the ground level.” Or again, to be absolutely precise, “at temperatures below T0, if the energy is measured then it is very likely that many of the building blocks from which the resulting energy eigenfunction is built are the ground level.” Read again the previous paragraph—the one that begins “What does all this mean physically?”. Notice that I never need to say that a particle “is in” or “goes into” a given level.
6.7.2 Experiment
References: M.H. Anderson, J.R. Ensher, M.R. Matthews, C.E. Wieman, E.A. Cornell, “Observation of Bose-Einstein condensation in a dilute atomic vapor”, Science, 269 (14 July 1995) 198–201; Randall G. Hullet at Rice; Malcolm W. Browne, “Two groups of physicists produce matter that Einstein postulated”, New York Times, 14 July 1995, page 1.
6.7.3 Problems
6.23 Character of the Bose function
What are the limits of the Bose function b(E) (equation 6.70) as E → ±∞? Is the curvature of the function greater when the temperature is high or when it is low?
6.24 Thermodynamics of the Bose condensate
For temperatures less than the Bose condensation temperature T0, find the energy, heat capacity, and entropy of an ideal gas of spin-zero bosons confined to a volume V. Write your answers in terms of the dimensionless integral
I=∫∞0x3/2ex−1dx
but don't bother to evaluate it. Show that
CV=52ET and S=53ET.
6.25 More thermodynamics of the Bose condensate
For the system of the previous problem, show that
F=−23E and p=23EV.
From this show that at low temperatures, the pressure of a collection of free and independent bosons goes like p ∼ T5/2. (This is always less than the classical pressure p ∼ T.)
6.26 An integral important for Bose condensation
Show that
∫∞0x1/2ex−1dx=ζ(32)√π2
where
ζ(s)=∞∑n=11ns.
Clue:
1ex−1=1ex(1−e−x)=e−x∞∑n=0e−nx for x>0.

