8.10: Electric Dipole Approximation
( \newcommand{\kernel}{\mathrm{null}\,}\)
In general, the wavelength of the type of electromagnetic radiation that induces, or is emitted during, transitions between different atomic energy levels is much larger than the typical size of a light atom. Thus,
exp[i(ω/c)n⋅x]=1+iωcn⋅x+⋯, ???can be approximated by its first term, unity (remember that ω/c=2π/λ ). This approximation is known as the electric dipole approximation. It follows that
⟨n|exp[i(ω/c)n⋅x] \( \mbox{\boldmath\)⋅p|i⟩≃ \( \mbox{\boldmath\)⋅⟨n|p|i⟩. ???It is readily demonstrated that
\( [{\bf x}, H_0] = \fracCallstack:
at (Under_Construction/Purgatory/Book:_Quantum_Mechanics_(Fitzpatrick)/08:_Time-Dependent_Perturbation_Theory/8.10:_Electric_Dipole_Approximation), /content/body/tr[3]/td[1]/span, line 1, column 1
so
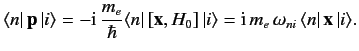 ???
??? Thus, making use of the electric dipole approximation, we obtain
wi→n=4π2αcℏu(ωni)|\( \mbox{\boldmath\)⋅fni|2 ???for absorption, and
wi→n=4π2αcℏu(ωin)|\( \mbox{\boldmath\)⋅fin|2 ???for stimulated emission, where
fni=⟨n|x|i⟩, ???and α=e2/(2ϵ0hc)≃1/137 is the fine structure constant.
Suppose that the radiation is polarized in the z -direction, so that ϵ =ez . We have already seen, from Section 7.4, that ⟨n|z|i⟩=0 unless the initial and final states satisfy
Δl =±1, ??? Δm =0. ???Here, l is the quantum number describing the total orbital angular momentum of the electron, and m is the quantum number describing the projection of the orbital angular momentum along the z -axis. It is easily demonstrated that ⟨n|x|i⟩ and ⟨n|y|i⟩ are only non-zero if
Δl =±1, ??? Δm =±1. ???Thus, for generally directed radiation ϵ ⋅fni is only non-zero if
Δl =±1, ??? Δm =0,±1. ???These are termed the selection rules for electric dipole transitions. It is clear, for instance, that the electric dipole approximation allows a transition from a 2p state to a 1s state, but disallows a transition from a 2s to a 1s state. The latter transition is called a forbidden transition.
Forbidden transitions are not strictly forbidden. Instead, they take place at a far lower rate than transitions that are allowed according to the electric dipole approximation. After electric dipole transitions, the next most likely type of transition is a magnetic dipole transition, which is due to the interaction between the electron spin and the oscillating magnetic field of the incident electromagnetic radiation. Magnetic dipole transitions are typically about 105 times more unlikely than similar electric dipole transitions. The first-order term in Equation ??? yields so-called electric quadrupole transitions. These are typically about 108 times more unlikely than electric dipole transitions. Magnetic dipole and electric quadrupole transitions satisfy different selection rules than electric dipole transitions. For instance, the selection rules for electric quadrupole transitions are Δl=0,±2 . Thus, transitions that are forbidden as electric dipole transitions may well be allowed as magnetic dipole or electric quadrupole transitions.
Contributors
Richard Fitzpatrick (Professor of Physics, The University of Texas at Austin)


