11.9: Zeeman Effect
( \newcommand{\kernel}{\mathrm{null}\,}\)
Consider a hydrogen atom placed in a uniform z-directed external magnetic field of magnitude |B|. The modification to the Hamiltonian of the system is H1=−μ⋅B, where μ=−e2me(L+2S) is the total electron magnetic moment, including both orbital and spin contributions. [See Equations ([e10.57])–([e10.59]).] Thus, H1=eB2me(Lz+2Sz).
Suppose that the applied magnetic field is much weaker than the atom’s internal magnetic field, ([e12.124]). Because the magnitude of the internal field is about 25 tesla, this is a fairly reasonable assumption. In this situation, we can treat H1 as a small perturbation acting on the simultaneous eigenstates of the unperturbed Hamiltonian and the fine structure Hamiltonian. Of course, these states are the simultaneous eigenstates of L2, S2, J2, and Jz. (See the previous section.) Hence, from standard perturbation theory, the first-order energy-shift induced by a weak external magnetic field is ΔEl,1/2;j,mj=⟨l,1/2;j,mj|H1|l,1/2;j,mj⟩=eB2me(mjℏ+⟨l,1/2;j,mj|Sz|l,1/2;j,mj⟩), because Jz=Lz+Sz. Now, according to Equations ([e11.47]) and ([e11.48]),
ψ(2)j,mj=(j+mj2l+1)1/2ψ(1)mj−1/2,1/2+(j−mj2l+1)1/2ψ(1)mj+1/2,−1/2 when j=l+1/2, and ψ(2)j,mj=(j+1−mj2l+1)1/2ψ(1)mj−1/2,1/2−(j+1+mj2l+1)1/2ψ(1)mj+1/2,−1/2 when j=l−1/2. Here, the ψ(1)m,ms are the simultaneous eigenstates of L2, S2, Lz, and Sz, whereas the ψ(2)j,mj are the simultaneous eigenstates of L2, S2, J2, and Jz. In particular, Szψ(1)m,±1/2=±ℏ2ψ(1)m,±1/2. It follows from Equations ([e12.143])–([e12.145]), and the orthormality of the ψ(1), that ⟨l,1/2;j,mj|Sz|l,1/2;j,mj⟩=±mjℏ2l+1 when j=l±1/2. Thus, the induced energy-shift when a hydrogen atom is placed in an external magnetic field—which is known as the Zeeman effect —becomes ΔEl,1/2;j,mj=μBBmj(1±12l+1) where the ± signs correspond to j=l±1/2. Here, μB=eℏ2me=5.788×10−5eV/T is known as the Bohr magnetron. Of course, the quantum number mj takes values differing by unity in the range −j to j. It, thus, follows from Equation ([e12.147]) that the Zeeman effect splits degenerate states characterized by j=l+1/2 into 2j+1 equally spaced states of interstate spacing ΔEj=l+1/2=μBB(2l+22l+1). Likewise, the Zeeman effect splits degenerate states characterized by j=l−1/2 into 2j+1 equally spaced states of interstate spacing ΔEj=l−1/2=μBB(2l2l+1).
In conclusion, in the presence of a weak external magnetic field, the two degenerate 1S1/2 states of the hydrogen atom are split by 2μBB. Likewise, the four degenerate 2S1/2 and 2P1/2 states are split by (2/3)μBB, whereas the four degenerate 2P3/2 states are split by (4/3)μBB. This is illustrated in Figure [fzee]. Note, finally, that because the ψ(2)l,mj are not simultaneous eigenstates of the unperturbed and perturbing Hamiltonians, Equations ([e12.149]) and ([e12.150]) can only be regarded as the expectation values of the magnetic-field induced energy-shifts. However, as long as the external magnetic field is much weaker than the internal magnetic field, these expectation values are almost identical to the actual measured values of the energy-shifts.
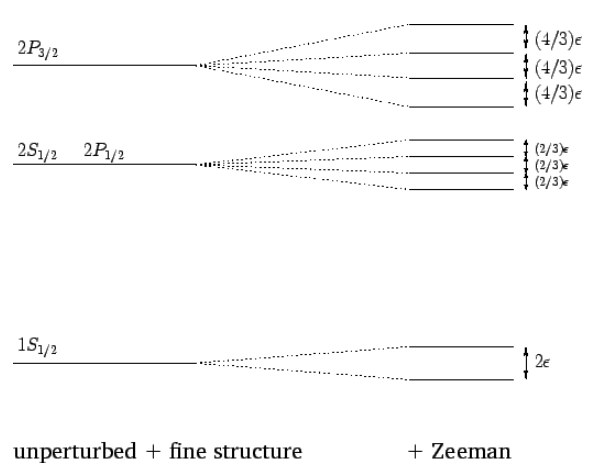
Figure 24: The Zeeman effect for the ![]() and
and ![]() states of a hydrogen atom. Here, ϵ=μBB. Not to scale.
states of a hydrogen atom. Here, ϵ=μBB. Not to scale.
Contributors and Attributions
Richard Fitzpatrick (Professor of Physics, The University of Texas at Austin)


