10.3: Nuclear Binding Energy
( \newcommand{\kernel}{\mathrm{null}\,}\)
By the end of this section, you will be able to:
- Calculate the mass defect and binding energy for a wide range of nuclei
- Use a graph of binding energy per nucleon (BEN) versus mass number (A) graph to assess the relative stability of a nucleus
- Compare the binding energy of a nucleon in a nucleus to the ionization energy of an electron in an atom
The forces that bind nucleons together in an atomic nucleus are much greater than those that bind an electron to an atom through electrostatic attraction. This is evident by the relative sizes of the atomic nucleus and the atom (10−15 and 10−10m, respectively). The energy required to pry a nucleon from the nucleus is therefore much larger than that required to remove (or ionize) an electron in an atom. In general, all nuclear changes involve large amounts of energy per particle undergoing the reaction. This has numerous practical applications.
Mass Defect
According to nuclear particle experiments, the total mass of a nucleus (mnuc) is less than the sum of the masses of its constituent nucleons (protons and neutrons). The mass difference, or mass defect, is given by
Δm=Zmp+(A−Z)mn−mnuc
where Zmp is the total mass of the protons, (A−Z)mn is the total mass of the neutrons, and mnuc is the mass of the nucleus. According to Einstein’s special theory of relativity, mass is a measure of the total energy of a system (E=mc2). Thus, the total energy of a nucleus is less than the sum of the energies of its constituent nucleons. The formation of a nucleus from a system of isolated protons and neutrons is therefore an exothermic reaction—meaning that it releases energy. The energy emitted, or radiated, in this process is (Δm)c2.
Now imagine this process occurs in reverse. Instead of forming a nucleus, energy is put into the system to break apart the nucleus (Figure 10.3.1). The amount of energy required is called the total binding energy (BE), Eb.
The binding energy is equal to the amount of energy released in forming the nucleus, and is therefore given by
Eb=(Δm)c2.
Experimental results indicate that the binding energy for a nucleus with mass number A>8 is roughly proportional to the total number of nucleons in the nucleus, A. The binding energy of a magnesium nucleus (24Mg), for example, is approximately two times greater than for the carbon nucleus (12C).
Calculate the mass defect and the binding energy of the deuteron. The mass of the deuteron is mD=3.34359×10−27kg or 1875.61MeV/c2.
Solution
For the deuteron Z=1 and A=2. From Equation ???, the mass defect for the deuteron is
Δm=mp+mn−mD=938.28MeV/c2+939.57MeV/c2−1875.61MeV/c2=2.24MeV/c2.
The binding energy of the deuteron is then
Eb=(Δm)c2=(2.24MeV/c2)(c2)=2.24MeV.
Over two million electron volts are needed to break apart a deuteron into a proton and a neutron. This very large value indicates the great strength of the nuclear force. By comparison, the greatest amount of energy required to liberate an electron bound to a hydrogen atom by an attractive Coulomb force (an electromagnetic force) is about 10 eV.
Graph of Binding Energy per Nucleon
In nuclear physics, one of the most important experimental quantities is the binding energy per nucleon (BEN), which is defined by
BEN=EbA
This quantity is the average energy required to remove an individual nucleon from a nucleus—analogous to the ionization energy of an electron in an atom. If the BEN is relatively large, the nucleus is relatively stable. BEN values are estimated from nuclear scattering experiments.
A graph of binding energy per nucleon versus atomic number A is given in Figure 10.3.2. This graph is considered by many physicists to be one of the most important graphs in physics. Two notes are in order. First, typical BEN values range from 6–10 MeV, with an average value of about 8 MeV. In other words, it takes several million electron volts to pry a nucleon from a typical nucleus, as compared to just 13.6 eV to ionize an electron in the ground state of hydrogen. This is why nuclear force is referred to as the “strong” nuclear force.
Second, the graph rises at low A, peaks very near iron (Fe,A=56), and then tapers off at high A. The peak value suggests that the iron nucleus is the most stable nucleus in nature (it is also why nuclear fusion in the cores of stars ends with Fe). The reason the graph rises and tapers off has to do with competing forces in the nucleus. At low values of A, attractive nuclear forces between nucleons dominate over repulsive electrostatic forces between protons. But at high values of A, repulsive electrostatic forces between forces begin to dominate, and these forces tend to break apart the nucleus rather than hold it together.
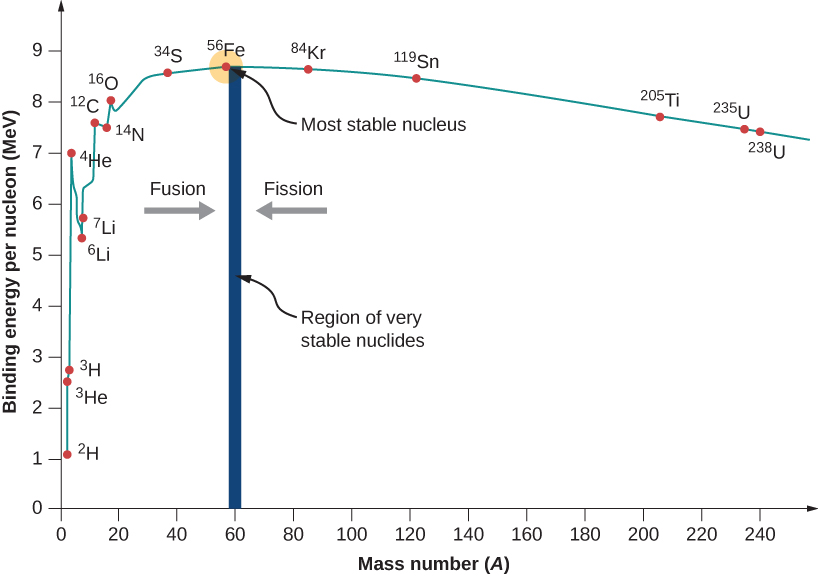
As we will see, the BEN-versus-A graph implies that nuclei divided or combined release an enormous amount of energy. This is the basis for a wide range of phenomena, from the production of electricity at a nuclear power plant to sunlight.
Calculate the binding energy per nucleon of an 4He(αparticle).
Strategy
Determine the total binding energy (BE) using the equation BE=(Δm)c2, where Δm is the mass defect. The binding energy per nucleon (BEN) is BE divided by A (Equation ???).
Solution
For 4He, we have Z=N=2. The total binding energy (Equation ???) is
BE=[2mp+2mn]−m(4He)c2.
These masses are m(4He)=4.002602u, mp=1.007825u, and mn=1.008665u. Thus we have
BE=(0.030378u)c2.
Noting that 1u=931.5MeV/c2, we find
BE=(0.030378)(931.5MeV/c2)c2=28.3MeV.
Since A=4, the total binding energy per nucleon (Equation ???) is
BEN=7.07MeV/nucleon.
Significance
Notice that the binding energy per nucleon for 4He is much greater than for the hydrogen isotopes (≈3MeV/nucleon). Therefore, helium nuclei cannot break down hydrogen isotopes without energy being put into the system.
If the binding energy per nucleon is large, does this make it harder or easier to strip off a nucleon from a nucleus?
- Answer
-
harder


