34.4: Procedures
( \newcommand{\kernel}{\mathrm{null}\,}\)
You will view the atomic spectra of various elements.
Warnings
- Do not touch the high voltage connections on the Spectrum Tube Power Supply
- Gas emission tubes become very hot and can burn the hand if touched
- Overflow light from electronic devices may inhibit observations
-
Start a list in which to record the element, the dominant color, and the pattern of colored lines (atomic spectrum). See example. You may either label the lines or use colored pencils to show the colors.
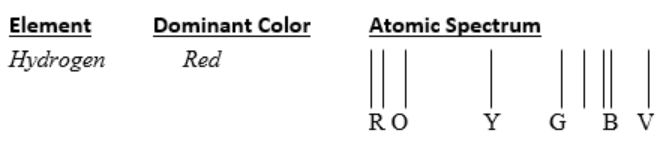
-
Make sure the voltage source is off. Place a gas emission tube into the voltage source. Turn on the voltage source and observe the dominant color, the color of the tube you see with your eyes, emitted from the center of the emission tube. Record the element contained in the gas tube and the dominant color you observed. Each person may interpret the dominant color slightly differently (i.e., pink vs red, vs orange).
-
When the lights are turned off, view the atomic spectrum produced through either the diffraction grating slide or the hand-held spectrometer. You may want to alternate devices and determine which the better tool is for you. Observe and remember the pattern of colored lines.
-
Once the lights are turned back on, sketch the pattern of colored lines, labeling each line or set of lines with the color. Attempt to show the relative spacing of the lines in your sketch. Your pattern could go from red to violet or violet to red, depending on how you are holding your observing device. Make sure you orient your observing device so the patterns have the same orientation for each viewing.
-
Observe all available elements and complete your list.

