11.2: Interstellar Gas
( \newcommand{\kernel}{\mathrm{null}\,}\)
Learning Objectives
By the end of this section, you will be able to:
- Name the major types of interstellar gas
- Discuss how we can observe each type
- Describe the temperature and other major properties of each type
Interstellar gas, depending on where it is located, can be as cold as a few degrees above absolute zero or as hot as a million degrees or more. We will begin our voyage through the interstellar medium by exploring the different conditions under which we find gas.
Ionized Hydrogen (H II) Regions—Gas Near Hot Stars
Some of the most spectacular astronomical photographs show interstellar gas located near hot stars (Figure 11.2.1). The strongest line in the visible region of the hydrogen spectrum is the red line in the Balmer series1 (as explained in the chapter on Radiation and Spectra); this emission line accounts for the characteristic red glow in images like Figure 11.2.1.

Hot stars are able to heat nearby gas to temperatures close to 10,000 K. The ultraviolet radiation from the stars also ionizes the hydrogen (remember that during ionization, the electron is stripped completely away from the proton). Such a detached proton won’t remain alone forever when attractive electrons are around; it will capture a free electron, becoming a neutral hydrogen once more. However, such a neutral atom can then absorb ultraviolet radiation again and start the cycle over. At a typical moment, most of the atoms near a hot star are in the ionized state.
Since hydrogen is the main constituent of interstellar gas, we often characterize a region of space according to whether its hydrogen is neutral or ionized. A cloud of ionized hydrogen is called an H II region. (Scientists who work with spectra use the Roman numeral I to indicate that an atom is neutral; successively higher Roman numerals are used for each higher stage of ionization. H II thus refers to hydrogen that has lost its one electron; Fe III is iron with two electrons missing.)
The electrons that are captured by the hydrogen nuclei cascade down through the various energy levels of the hydrogen atoms on their way to the lowest level, or ground state. During each transition downward, they give up energy in the form of light. The process of converting ultraviolet radiation into visible light is called fluorescence. Interstellar gas contains other elements besides hydrogen. Many of them are also ionized in the vicinity of hot stars; they then capture electrons and emit light, just as hydrogen does, allowing them to be observed by astronomers. But generally, the red hydrogen line is the strongest, and that is why H II regions look red.
A fluorescent light on Earth works using the same principles as a fluorescent H II region. When you turn on the current, electrons collide with atoms of mercury vapor in the tube. The mercury is excited to a high-energy state because of these collisions. When the electrons in the mercury atoms return to lower-energy levels, some of the energy they emit is in the form of ultraviolet photons. These, in turn, strike a phosphor-coated screen on the inner wall of the light tube. The atoms in the screen absorb the ultraviolet photons and emit visible light as they cascade downward among the energy levels. (The difference is that these atoms give off a wider range of light colors, which mix to give the characteristic white glow of fluorescent lights, whereas the hydrogen atoms in an H II region give off a more limited set of colors.)
Neutral Hydrogen Clouds
The very hot stars required to produce H II regions are rare, and only a small fraction of interstellar matter is close enough to such hot stars to be ionized by them. Most of the volume of the interstellar medium is filled with neutral (nonionized) hydrogen. How do we go about looking for it?
Unfortunately, neutral hydrogen atoms at temperatures typical of the gas in interstellar space neither emit nor absorb light in the visible part of the spectrum. Nor, for the most part, do the other trace elements that are mixed with the interstellar hydrogen. However, some of these other elements can absorb visible light even at typical interstellar temperatures. This means that when we observe a bright source such as a hot star or a galaxy, we can sometimes see additional lines in its spectrum produced when interstellar gas absorbs light at particular frequencies (see Figure). Some of the strongest interstellar absorption lines are produced by calcium and sodium, but many other elements can be detected as well in sufficiently sensitive observations (as discussed in Radiation and Spectra).
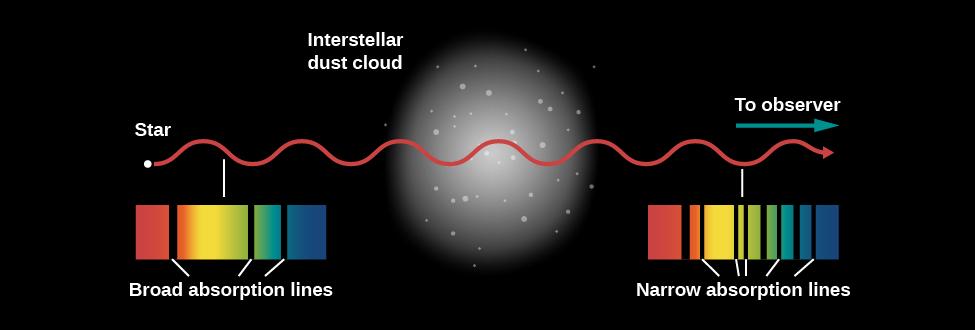
The first evidence for absorption by interstellar clouds came from the analysis of a spectroscopic binary star (see The Stars: A Celestial Census), published in 1904. While most of the lines in the spectrum of this binary shifted alternately from longer to shorter wavelengths and back again, as we would expect from the Doppler effect for stars in orbit around each other, a few lines in the spectrum remained fixed in wavelength. Since both stars are moving in a binary system, lines that showed no motion puzzled astronomers. The lines were also peculiar in that they were much, much narrower than the rest of the lines, indicating that the gas producing them was at a very low pressure. Subsequent work demonstrated that these lines were not formed in the star’s atmosphere at all, but rather in a cold cloud of gas located between Earth and the binary star.
While these and similar observations proved there was interstellar gas, they could not yet detect hydrogen, the most common element, due to its lack of spectral features in the visible part of the spectrum. (The Balmer line of hydrogen is in the visible range, but only excited hydrogen atoms produce it. In the cold interstellar medium, the hydrogen atoms are all in the ground state and no electrons are in the higher-energy levels required to produce either emission or absorption lines in the Balmer series.) Direct detection of hydrogen had to await the development of telescopes capable of seeing very-low-energy changes in hydrogen atoms in other parts of the spectrum. The first such observations were made using radio telescopes, and radio emission and absorption by interstellar hydrogen remains one of our main tools for studying the vast amounts of cold hydrogen in the universe to this day.
In 1944, while he was still a student, the Dutch astronomer Hendrik van de Hulst predicted that hydrogen would produce a strong line at a wavelength of 21 centimeters. That’s quite a long wavelength, implying that the wave has such a low frequency and low energy that it cannot come from electrons jumping between energy levels (as we discussed in Radiation and Spectra). Instead, energy is emitted when the electron does a flip, something like an acrobat in a circus flipping upright after standing on his head.
The flip works like this: a hydrogen atom consists of a proton and an electron bound together. Both the proton and the electron act is if they were spinning like tops, and spin axes of the two tops can either be pointed in the same direction (aligned) or in opposite directions (anti-aligned). If the proton and electron were spinning in opposite directions, the atom as a whole would have a very slightly lower energy than if the two spins were aligned (Figure 11.2.3). If an atom in the lower-energy state (spins opposed) acquired a small amount of energy, then the spins of the proton and electron could be aligned, leaving the atom in a slightly excited state. If the atom then lost that same amount of energy again, it would return to its ground state. The amount of energy involved corresponds to a wave with a wavelength of 21 centimeters; hence, it is known as the 21-centimeter line.
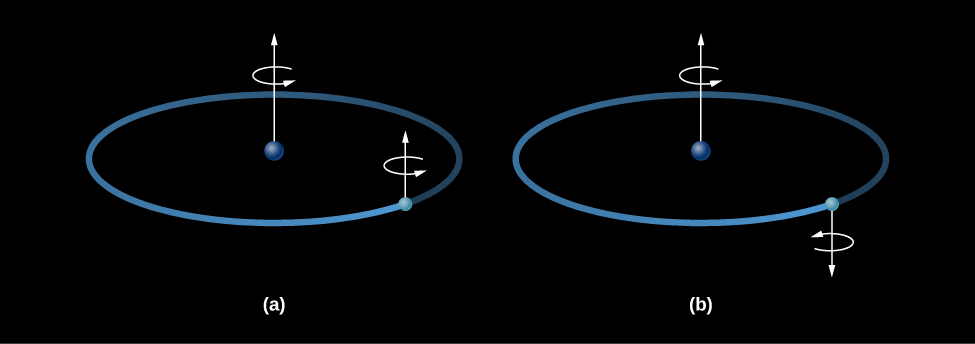
Neutral hydrogen atoms can acquire small amounts of energy through collisions with other hydrogen atoms or with free electrons. Such collisions are extremely rare in the sparse gases of interstellar space. An individual atom may wait centuries before such an encounter aligns the spins of its proton and electron. Nevertheless, over many millions of years, a significant fraction of the hydrogen atoms are excited by a collision. (Out there in cold space, that’s about as much excitement as an atom typically experiences.)
An excited atom can later lose its excess energy either by colliding with another particle or by giving off a radio wave with a wavelength of 21 centimeters. If there are no collisions, an excited hydrogen atom will wait an average of about 10 million years before emitting a photon and returning to its state of lowest energy. Even though the probability that any single atom will emit a photon is low, there are so many hydrogen atoms in a typical gas cloud that collectively they will produce an observable line at 21 centimeters.
Equipment sensitive enough to detect the 21-cm line of neutral hydrogen became available in 1951. Dutch astronomers had built an instrument to detect the 21-cm waves that they had predicted, but a fire destroyed it. As a result, two Harvard physicists, Harold Ewen and Edward Purcell, made the first detection (Figure 11.2.4), soon followed by confirmations from the Dutch and a group in Australia. Since the detection of the 21-cm line, many other radio lines produced by both atoms and molecules have been discovered (as we will discuss in a moment), and these have allowed astronomers to map out the neutral gas throughout our home Galaxy. Astronomers have also detected neutral interstellar gas, including hydrogen, at many other wavelengths from the infrared to the ultraviolet.

Modern radio observations show that most of the neutral hydrogen in our Galaxy is confined to an extremely flat layer, less than 300 light-years thick, that extends throughout the disk of the Milky Way Galaxy. This gas has densities ranging from about 0.1 to about 100 atoms per cm3 , and it exists at a wide range of temperatures, from as low as about 100 K (–173 °C) to as high as about 8000 K. These regions of warm and cold gas are interspersed with each other, and the density and temperature at any particular point in space are constantly changing.
Ultra-Hot Interstellar Gas
While the temperatures of 10,000 K found in H II regions might seem warm, they are not the hottest phase of the interstellar medium. Some of the interstellar gas is at a temperature of a million degrees, even though there is no visible source of heat nearby. The discovery of this ultra-hot interstellar gas was a big surprise. Before the launch of astronomical observatories into space, which could see radiation in the ultraviolet and X-ray parts of the spectrum, astronomers assumed that most of the region between stars was filled with hydrogen at temperatures no warmer than those found in H II regions. But telescopes launched above Earth’s atmosphere obtained ultraviolet spectra that contained interstellar lines produced by oxygen atoms that have been ionized five times. To strip five electrons from their orbits around an oxygen nucleus requires a lot of energy. Subsequent observations with orbiting X-ray telescopes revealed that the Galaxy is filled with numerous bubbles of X-ray-emitting gas. To emit X-rays, and to contain oxygen atoms that have been ionized five times, gas must be heated to temperatures of a million degrees or more.
Theorists have now shown that the source of energy producing these remarkable temperatures is the explosion of massive stars at the ends of their lives (Figure 11.2.5). Such explosions, called supernovae, will be discussed in detail in the chapter on The Death of Stars. For now, we’ll just say that some stars, nearing the ends of their lives, become unstable and literally explode. These explosions launch gas into interstellar space at velocities of tens of thousands of kilometers per second (up to about 30% the speed of light). When this ejected gas collides with interstellar gas, it produces shocks that heat the gas to millions or tens of millions of degrees.

Astronomers estimate that one supernova explodes roughly every 100 years somewhere in the Galaxy. On average, shocks launched by supernovae sweep through any given point in the Galaxy about once every few million years. These shocks keep some interstellar space filled with gas at temperatures of millions of degrees, and they continually disturb the colder gas, keeping it in constant, turbulent motion.
Molecular Clouds
A few simple molecules out in space, such as CN and CH, were discovered decades ago because they produce absorption lines in the visible-light spectra of stars behind them. When more sophisticated equipment for obtaining spectra in radio and infrared wavelengths became available, astronomers—to their surprise—found much more complex molecules in interstellar clouds as well.
Just as atoms leave their “fingerprints” in the spectrum of visible light, so the vibration and rotation of atoms within molecules can leave spectral fingerprints in radio and infrared waves. If we spread out the radiation at such longer wavelengths, we can detect emission or absorption lines in the spectra that are characteristic of specific molecules. Over the years, experiments in our laboratories have shown us the exact wavelengths associated with changes in the rotation and vibration of many common molecules, giving us a template of possible lines against which we can now compare our observations of interstellar matter.
The discovery of complex molecules in space came as a surprise because most of interstellar space is filled with ultraviolet light from stars, and this light is capable of dissociating molecules (breaking them apart into individual atoms). In retrospect, however, the presence of molecules is not surprising. As we will discuss further in the next section, and have already seen above, interstellar space also contains significant amounts of dust capable of blocking out starlight. When this dust accumulates in a single location, the result is a dark cloud where ultraviolet starlight is blocked and molecules can survive. The largest of these structures are created where gravity pulls interstellar gas together to form giant molecular clouds, structures as massive as a million times the mass of the Sun. Within these, most of the interstellar hydrogen has formed the molecule H2 (molecular hydrogen). Other, more complex molecules are also present in much smaller quantities.
Giant molecular clouds have densities of hundreds to thousands of atoms per cm3, much denser than interstellar space is on average. As a result, though they account for a very small fraction of the volume of interstellar space, they contain a significant fraction—20–30%—of the total mass of the Milky Way’s gas. Because of their high density, molecular clouds block ultraviolet starlight, the main agent for heating most interstellar gas. As a result, they tend to be extremely cold, with typical temperatures near 10 K (−263 °C). Giant molecular clouds are also the sites where new stars form, as we will discuss below.
It is in these dark regions of space, protected from starlight, that molecules can form. Chemical reactions occurring both in the gas and on the surface of dust grains lead to much more complex compounds, hundreds of which have been identified in interstellar space. Among the simplest of these are water (H2O), carbon monoxide (CO), which is produced by fires on Earth, and ammonia (NH3), whose smell you recognize in strong home cleaning products. Carbon monoxide is particularly abundant in interstellar space and is the primary tool that astronomers use to study giant molecular clouds. Unfortunately, the most abundant molecule, H2, is particularly difficult to observe directly because in most giant molecular clouds, it is too cold to emit even at radio wavelengths. CO, which tends to be present wherever H2 is found, is a much better emitter and is often used by astronomers to trace molecular hydrogen.
The more complex molecules astronomers have found are mostly combinations of hydrogen, oxygen, carbon, nitrogen, and sulfur atoms. Many of these molecules are organic (those that contain carbon and are associated with the carbon chemistry of life on Earth.) They include formaldehyde (used to preserve living tissues), alcohol (see the feature box on Cocktails in Space below), and antifreeze.
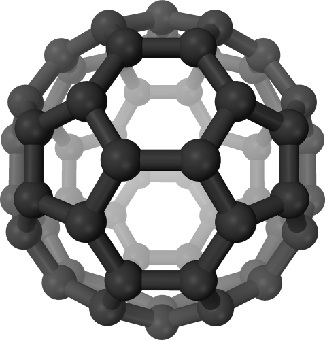
In 1996, astronomers discovered acetic acid (the prime ingredient of vinegar) in a cloud lying in the direction of the constellation of Sagittarius. To balance the sour with the sweet, a simple sugar (glycolaldehyde) has also been found. The largest compounds yet discovered in interstellar space are fullerenes, molecules in which 60 or 70 carbon atoms are arranged in a cage-like configuration (see Figure 11.2.6). See Table 11.2.1 below for a list of a few of the more interesting interstellar molecules that have been found so far.
| Table 11.2.1: Some Interesting Interstellar Molecules | ||
|---|---|---|
| Name | Chemical Formula | Use on Earth |
| Ammonia | NH3 | Household cleansers |
| Formaldehyde | H2CO | Embalming fluid |
| Acetylene | HC2H | Fuel for a welding torch |
| Acetic acid | C2H2O4 | The essence of vinegar |
| Ethyl alcohol | CH3CH2OH | End-of-semester parties |
| Ethylene glycol | HOCH2CH2OH | Antifreeze ingredient |
| Benzene | C6H6 | Carbon ring, ingredient in varnishes and dyes |
The cold interstellar clouds also contain cyanoacetylene (HC3N) and acetaldehyde (CH3CHO), generally regarded as starting points for amino acid formation. These are building blocks of proteins, which are among the fundamental chemicals from which living organisms on Earth are constructed. The presence of these organic molecules does not imply that life exists in space, but it does show that the chemical building blocks of life can form under a wide range of conditions in the universe. As we learn more about how complex molecules are produced in interstellar clouds, we gain an increased understanding of the kinds of processes that preceded the beginnings of life on Earth billions of years ago.
Interested in learning more about fullerenes, buckyballs, or buckminsterfullerenes (as they’re called)? Watch a brief video from NASA’s Jet Propulsion Laboratory that explains what they are and illustrates how they were discovered in space.
COCKTAILS IN SPACE
Among the molecules astronomers have identified in interstellar clouds is alcohol, which comes in two varieties: methyl (or wood) alcohol and ethyl alcohol (the kind you find in cocktails). Ethyl alcohol is a pretty complex molecule, written by chemists as C2H5OH. It is quite plentiful in space (relatively speaking). In clouds where it has been identified, we detect up to one molecule for every m3. The largest of the clouds (which can be several hundred light-years across) have enough ethyl alcohol to make 1028 fifths of liquor.
We need not fear, however, that future interstellar astronauts will become interstellar alcoholics. Even if a spaceship were equipped with a giant funnel 1 kilometer across and could scoop it through such a cloud at the speed of light, it would take about a thousand years to gather up enough alcohol for one standard martini.
Furthermore, the very same clouds also contain water (H2O) molecules. Your scoop would gather them up as well, and there are a lot more of them because they are simpler and thus easier to form. For the fun of it, one astronomical paper actually calculated the proof of a typical cloud. Proof is the ratio of alcohol to water in a drink, where 0 proof means all water, 100 proof means half alcohol and half water, and 200 proof means all alcohol. The proof of the interstellar cloud was only 0.2, not enough to qualify as a stiff drink
Summary
Interstellar gas may be hot or cold. Gas found near hot stars emits light by fluorescence, that is, light is emitted when an electron is captured by an ion and cascades down to lower-energy levels. Glowing clouds (nebulae) of ionized hydrogen are called H II regions and have temperatures of about 10,000 K. Most hydrogen in interstellar space is not ionized and can best be studied by radio measurements of the 21-centimeter line. Some of the gas in interstellar space is at a temperature of a million degrees, even though it is far away in hot stars; this ultra-hot gas is probably heated when rapidly moving gas ejected in supernova explosions sweeps through space. In some places, gravity gathers interstellar gas into giant clouds, within which the gas is protected from starlight and can form molecules; more than 200 different molecules have been found in space, including the basic building blocks of proteins, which are fundamental to life as we know it here on Earth.
Footnotes
1Scientists also call this red Balmer line the H-alpha line, with alpha meaning it is the first spectral line in the Balmer series.
Glossary
- molecular cloud
- a large, dense, cold interstellar cloud; because of its size and density, this type of cloud can keep ultraviolet radiation from reaching its interior, where molecules are able to form
- H II region
- the region of ionized hydrogen in interstellar space
Contributors and Attributions
Andrew Fraknoi (Foothill College), David Morrison (NASA Ames Research Center), Sidney C. Wolff (National Optical Astronomy Observatory) with many contributing authors. Textbook content produced by OpenStax College is licensed under a Creative Commons Attribution License 4.0 license. Download for free at https://openstax.org/details/books/astronomy).


