5.3: Heat Capacity and Phase Transitions
- Page ID
- 18468
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Definition of Heat
In the pantheon of misused physics terms, at the top of the list must be the word heat. Standard usage would have us believe that it is a quantity of energy stored within a system, measurable by temperature. But we already have a term for that – thermal energy. Heat is a much more subtle concept.
In Physics 9A, we learned about the idea of work as a means of transferring energy between systems, or between types (e.g. from potential energy to kinetic energy, or mechanical energy to thermal energy). Clearly it would make no sense to say that two systems contain a certain amount of work, which then can be transferred between them. The definition of work makes it clear that it only exists as a transfer – the contained energy is kinetic, potential, or thermal, and when energy is moved or converted through a force acting through a distance, the energy is transferred through the process of work.
Heat is precisely like work in this regard. It is a process of the transfer of energy between systems, which necessarily requires an interaction between the systems. In the case of work, the interaction involved is a macroscopic net force. The emphasis here needs to be on the word "net," which indicates an imbalance. For heat transfer to occur, there also needs to be an imbalance, but in that case the imbalance needs to be in temperature. Microscopically, this means that when two systems come into thermal contact, the random motions of particles naturally tend toward bringing the average energies of the particles in both systems into balance. In short, work is energy transfer resulting from a (macroscopic, non-random) force imbalance, while heat is energy transfer resulting from a temperature imbalance, which is expressed microscopically as an imbalance in average particle energy.
Heat Capacity
We know that work can convert energy back-and-forth between kinetic and potential energy (when the force is conservative), and can transfer energy into thermal form (when the force is non-conservative), but because heat involves temperature differences, it is obviously intimately tied to thermal energy, and not mechanical energy. The most natural first question to ask is therefore, "How is the amount of heat transferred into or out of a system related to the temperature change that results from that transfer?"
We can answer this question through direct experiment, and the results of such an experiment are quite intuitive. Given that the temperature is a measure of average particle energy, then it stands to reason that if we add a known quantity of heat to a sample and the temperature goes up (say) \(1^oC\), we would expect that adding the same amount of energy again to the same sample would have the same effect. After all, the energy added is divided among the same number of particles each time, so the average energy per particle will go up the same each time. We express this result as a statement of proportionality:
\[Q \propto \Delta T\]
Another fairly obvious factor is the amount of matter in the sample. Again, the added heat is distributed amongst the particles in the sample, and the temperature change is a measure of how much their average energies go up. If we put in a known quantity of heat, then the more particles there are in the sample to share this energy, the less each particle's temperature changes. This means that the heat needed to raise the temperature a certain amount is proportional to the amount of matter in the sample. Now the "amount of matter" can be measured in many ways, and we will use two different ones, depending upon which is more convenient. Here we will look at the most obvious choice – the mass of the sample. We therefore extend the proportionality above to a factor of the amount of mass:
\[Q \propto m\Delta T\]
It's clear that the units don't match here, so there will need to be a constant of proportionality to turn this into an equality. But there may be additional information that needs to be contained within that constant as well. In this case we really do need to appeal to experiment, and we find that different substances require different constants for the equality to hold. We therefore introduce the specific heat capacity, \(c\), which differs from one substance to the next, and which completes the relationship between heat transferred and temperature change:
The mass is always a positive quantity, and we define the specific heat capacity as a positive value, which means that \(Q>0\) when \(\Delta T>0\). The temperature of a system rises when heat enters it, so in defining heat capacity as a positive value, we set the following sign convention:
\(Q>0\) whenever heat enters a system, and \(Q<0\) whenever heat exits a system
Digression: Specific Heat Capacity
Perhaps the reader is wondering where the name for this constant comes from. The word "specific" generally comes into scientific vernacular whenever one wants to factor out the role of a sample's mass. For example, the "specific gravity" of a sample is the ratio of its mass to the mass of an equal volume of water. So two samples of the same substance that have different masses have the same specific gravity. The specific heat capacity factors out the role of mass as well – i.e. the heat capacity per unit mass.
The second question is how the word "capacity" applies. The idea is that the heat capacity (the product of specific heat capacity and mass) is a measure of a sample's "capacity" to take in heat energy for a given temperature increase. Samples with a large heat capacity can take in a lot of heat energy without changing their temperature very much. There is a similar use of a derivative of the word "capacity" (called "capacitance") in the field of electromagnetic theory, which you will study in Physics 9C. In this case, it measures a system's "capacity" to separate charge across a given voltage difference.
The mention of specific gravity in the digression points out a common theme in chemistry and physics – using water as a standard for units of measurement. We already have a standard set of units (SI) that we have been using since day 1 of Physics 9A, but it is useful to point out a very common unit of energy – the calorie. This is defined as the amount of heat energy that must be added to 1 gram of water to raise its temperature by 1 degree Celsius. In other words:
\[c_{water} = \dfrac{Q}{m\Delta T} = \dfrac{1\;calorie}{\left(1\;g\right)\left(1^oC\right)} \]
We can, of course, convert between calories and joules, and it turns out that:
\[ 1 cal = 4.184J \;\;\; \Rightarrow \;\;\; c_{liquid\;water} = 4184 \dfrac{J}{kg^oC} \]
Alert
Because calculations involving heat capacity involve changes in temperature, it's okay to use Celsius in the units rather than Kelvins – changes in one of these equals changes in the other.
This provides us with a powerful tool for solving problems. If we put a hot piece of metal into a container of cool water, we can relate the starting temperatures to the final temperature reached when the two substances come to equilibrium. All we need to do is invoke conservation of energy (the heat that leaves one substance enters the other – and be careful to follow sign conventions properly), and use the above expressions. We can also measure the change in temperature in a reservoir of water that surrounds a chemical reaction to determine how much energy goes into or comes out of a chemical reaction. This process is commonly referred to as calorimetry.
Example \(\PageIndex{1}\)
A \(50.0\;g\) chunk of aluminum is left in a pot of boiling water at \(100^oC\) until it comes to thermal equilibrium with the water. It is then placed into an insulated vessel containing \(200\;g\) of water at \(20.0^oC\), and the system is allowed to come to thermal equilibrium. Find the final temperature this system reaches. The specific heat capacity of aluminum is \(0.215 \dfrac{cal}{g \;^oC}\).
- Solution
-
The starting point is noting that the insulated container allows for no heat transfer to or from outside the system, which means that all heat exchanged is between the aluminum and the water. All the heat that leaves the hot aluminum enters the cool water, so energy conservation (and our sign convention for heat) demands (the "a" subscript refers to aluminum and the "w" to water):
\[Q_a = - Q_w \nonumber\]
Now plugging in Equation 5.3.3 for \(Q\), we have:
\[m_a c_a \Delta T_a = - m_w c_w \Delta T_w\nonumber\]
Now we use the fact that the changes in temperature go from the starting temperatures to the same final temperature (thermal equilibrium), and solve for the final temperature:
\[m_a c_a \left(T-T_a\right) = - m_w c_w \left(T-T_w\right) \;\;\;\Rightarrow\;\;\; T = \dfrac{m_ac_aT_a+m_wc_wT_w}{m_ac_a+m_wc_w} = 24.1^oC \nonumber\]
Note that the final expression before the numerical answer is exactly a weighted average of the two temperatures, with the weightings of each temperature being the heat capacity of each substance.
The second version of heat capacity we will work with changes the proportionality into terms of moles rather than mass. In this case, we refer to the constant as molar heat capacity, and we denote it with an upper-case \(C\):
The \(n\) in this formula is the number of moles in the sample. We will find this version to be very useful for gases in particular. It also fits well with some of the more general thermodynamic concepts that we will get to later.
Phase Transitions and Latent Heat
When it comes to heat transferring into or out of substances, changing the temperature of the sample is not all that can occur. The sample can also undergo a phase change. By "phase" we mean the solid, liquid, and gaseous states of matter. These differ primarily in how the particles involved interact with each other. Adding heat to a sample changes part or all of it from solid into liquid (melting), or from liquid into gas (boiling). Having heat exit a sample can result in the phase changing in the opposite direction: liquid to solid (freezing) or gas to liquid (condensing).
What is interesting about phase changes is that they occur at a fixed temperature. The temperature at which solid/liquid transitions occur is called the melting point of that substance, while the temperature at which liquid/gas transitions occur is called the boiling point. Note that the temperature of melting is the same as the temperature of freezing, and the temperature of boiling is the same as the temperature of condensing. A transitions that occurs at a fixed temperature requires a different relationship than above between the heat transferred and the change it invokes. Rather than being proportional to the amount of temperature change, the heat added/lost is proportional to the amount of mass made to change phase. We write it this way:
\[Q = \pm L\;\delta m \]
The value \(\delta m\) employs a lowercase \(\delta\) to express a the amount of mass that changes phase which is assumed to always be a positive quantity, without invoking the "after-minus-before" that one generally associates with use of the uppercase \(\Delta\) – the amount of mass present doesn't change (it doesn't go anywhere), there is just some amount of mass that is changing phase. The sign \(\pm\) is positive when heat is entering the system, \(Q>0\), and the substance is either melting or vaporizing. The sign is negative when heat is leaving the system, \(Q<0\), and the substance is either freezing or condensing. The constant \(L\) is called the latent heat. This is a rather confusing name for this quantity, on a couple of counts. First, it is not heat – it doesn't even have the units of energy. And second, "latent" seems to imply that there is some heat sitting around stored in a sample, a misconception we have already addressed above. For any given substance, the constant \(L\) is different if it involves a phase change between solid and liquid than if it involves liquid and gas. The constant associated with the former we call the latent heat of fusion (\(L_f\)), and the the constant that deals with the latter is called the latent heat of vaporization (\(L_v\)).
Naturally processes where heat is exchanged can involve both temperature change and phase change at the same time (such as ice melting in a cup of warm water until equilibrium is reached), but assuming the system where the process is occurring is insulated, energy conservation (heat lost by one substance is gained by the other) shows the way.
Example \(\PageIndex{2}\)
A sealed insulated vessel containing a quantity of water is heated until it is brought to a boil, and the heating continues until all of the water is steam at a temperature of \(245^oC\). One fifth as much boiling (liquid) water is then injected into the vessel and the contents are held at constant pressure and are allowed to come to equilibrium. Find the final state of the contents of the vessel – the percentage of water in each phase (if mixed), or the temperature of the contents (if all in one phase). The specific heat capacity (at constant pressure) of steam is about \(1900\;\frac{J}{kg^oC}\), and the latent heat of vaporization of water is \(2.256\times 10^6\;\frac{J}{kg}\).
- Solution
-
Let's start with a sketch to clarify what is happening here:
Energy is leaving the hotter steam and entering the cooler water. At least for awhile, this will result in the steam cooling off while some of the water changes phase to steam. We won’t know, until we do the calculation, if all of the water will change to steam (and then get hotter than the boiling point), or if the steam will come down to the boiling point, thereby reaching thermal equilibrium. We’ll start by calculating the energies required to do both of these things – unfortunately there is no shortcut to avoid having to do this.
\[\begin{array}{l} \text{cooling the steam to }100^oC: && Q_1=mc_s\Delta T = m\left(1900\;\frac{J}{kg^oC}\right)\left(-145^oC\right) = m\left(275,500\;\frac{J}{kg}\right) \\ \text{water to steam:} && Q_2=L_v \delta m = \left(\dfrac{m}{5}\right) \left(2.256\times 10^6\;\frac{J}{kg}\right) = m\left(451,200\;\frac{J}{kg}\right)\end{array} \nonumber \]
We see that it takes more energy to turn all of the liquid water into steam than to reduce the temperature of the steam to the boiling point, so this heat transfer will end with a mix of liquid water and steam at \(100^oC\). We now calculate the mass of the water changed to steam in terms of the starting mass of the steam, using energy conservation:
\[0 = Q_1 + Q_2 = -m\left(275,500\;\frac{J}{kg}\right) + \delta m \left(2.256\times 10^6\;\frac{J}{kg}\right) \;\;\;\Rightarrow\;\;\; \delta m = 0.122m\nonumber\]
The total amount of steam at the end can now be divided by the total amount of steam + liquid water to get the percentages:
\[\left. \begin{array}{l} m_{steam} = m+\delta m = 1.122m \\ m_{total} = m + \dfrac{m}{5} \end{array} \right\} \dfrac{m_{steam}}{m_{total}} = 0.935 \nonumber \]
So the final result is 93.5% steam and 6.5% liquid water, at a temperature of \(100^oC\).
Suppose we wanted to transition a very cold solid all the way to a very hot gas. There would be several steps involved:
- add energy to raise the solid's temperature to the melting point
- add energy to change the phase from solid to liquid (while not changing the temperature)
- add energy to raise the liquid's temperature to the boiling point
- add energy to change the phase from liquid to gas (while not changing the temperature)
- add energy to raise the gas's temperature
Every one of these steps involves a different constant. The specific heat capacities of the solid, liquid, and gaseous phases of the same substance are not the same, and the latent heat of fusion is not the same as the latent heat of vaporization for the same substance. The full process is depicted in the graph below.
Figure 5.3.1 – Effect of Heat Transfer on Temperature and Phase
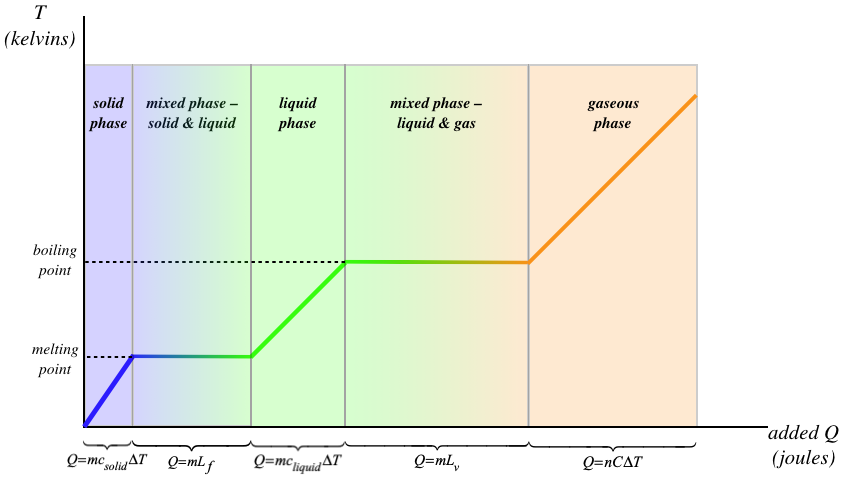
[Note: In the final section of the graph, you'll note that the molar heat capacity is used. This is because it is more common to measure the amount of a gas in moles rather than in units of mass.]


