8.1: Modeling Semiconductors (Project)
( \newcommand{\kernel}{\mathrm{null}\,}\)
I. Conductors, Semiconductors and Insulators
In a gas, where inter-atomic distances are large, the energy levels of one gas atom are not altered by the presence of the other gas atoms. Thus, you can think of each atom as a separate entity. A simplified energy level diagram for two gas atoms would look like this:
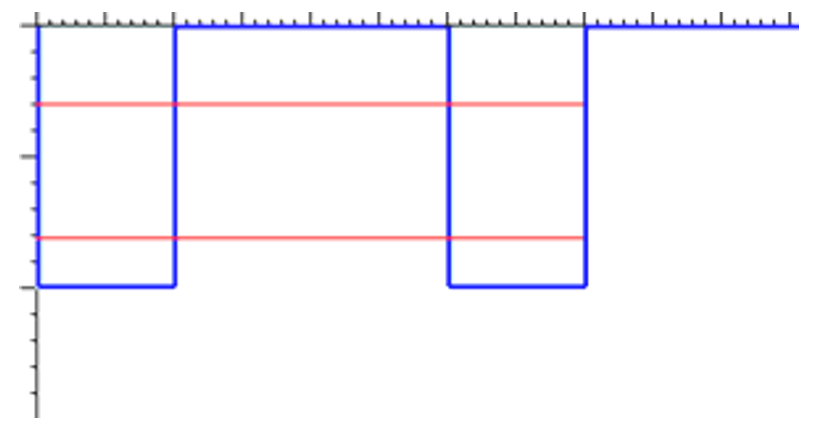
However, in a solid the atoms are close enough together that they directly influence each other’s energy levels. For example, in a hypothetical two-atom solid (a pretty small solid), the energy levels look like this:
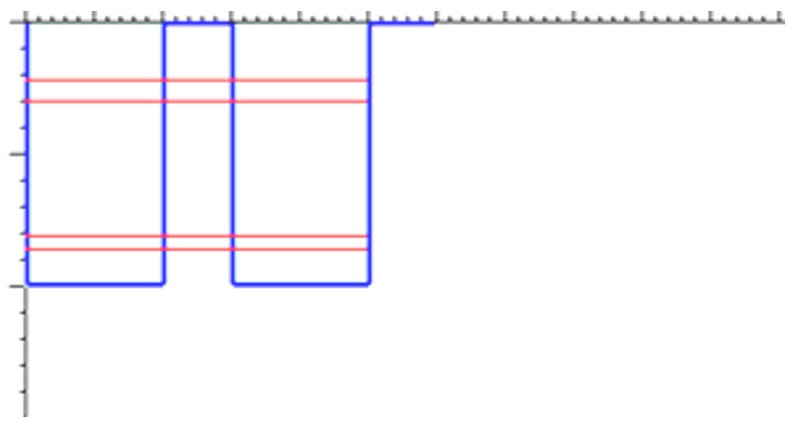
The splitting in the energy levels occurs because an “odd” wavefunction (positive in one well and negative in the other) has a different energy than an “even” wavefunction (positive in both wells).
If we were to look at realistic solids (with more than just a handful of atoms), this discreet splitting into two levels becomes a continuous blurring of energy levels into energy bands.
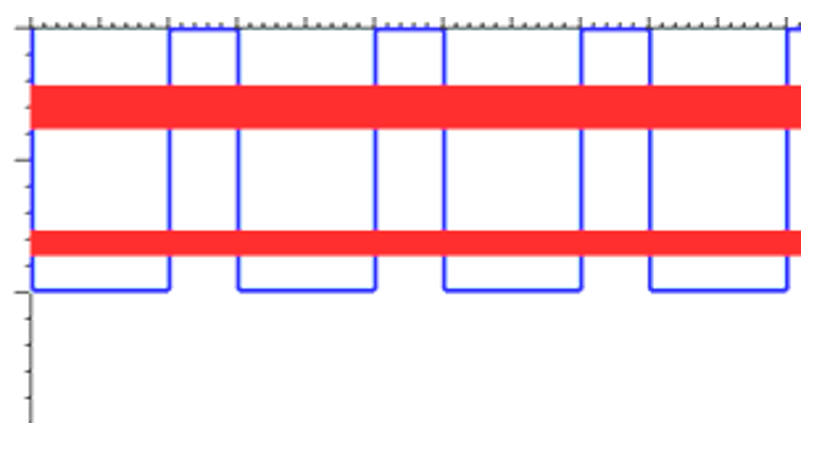
Thus, the allowed energy levels for electrons in solids are actually continuous bands separated by finite gaps in energy. The size of the band gap between the highest filled energy band (the valence band) and the lowest unfilled energy band (the conduction band) is of crucial importance in determining the solids properties.
For example, if this band gap is large (a few eV), electrons in the valence band will not be able to easily “jump” into the conduction band and the solid will be an electrical insulator. If the gap is basically non-existent, the electrons have free access to the conduction band and the solid is a conductor. If the gap is small (near 1 eV), the solid has a rich variety of “intermediate” properties that make into particularly intriguing. These materials are intrinsic semiconductors. The three possibilities are illustrated below.
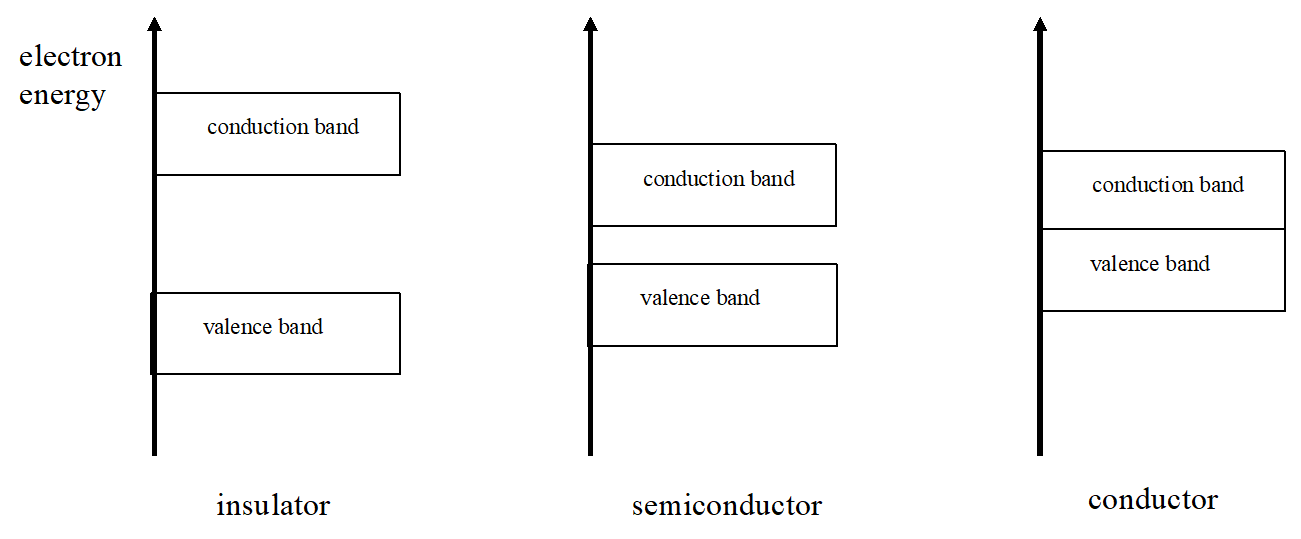
II. Intrinsic Semiconductors
Imagine an electron in the valence band of a semiconductor. This electron is bound to its “parent” atom, but longs to be free to roam the entire solid. If only it could “jump” up to the conduction band! If it could, it would no longer be bound to any one atom but rather it would be free to flow wherever forces take it in the solid. In other words, it could be part of a current. But where will it get the energy needed to make this jump? From the random thermal vibrations of the solid!
In general, the relative probability of acquiring an amount of energy, E, from the thermal energy available in a material is given by the Boltzmann Factor:
Probability≈e−E/kT
where E is the energy acquired, T is the absolute temperature, and k is Boltzmann’s constant (8.617×10−5eVK−1).
For quantum mechanical reasons we’ll gloss over, the effective energy needed to jump the gap is only about ½ of the actual band gap (and even this factor of ½ depends on the precise manufacturing of the semiconductor). Thus, the probability of an electron jumping from the valence band to the conduction band is:
Probability≈e−Egap/2kT
where Egap is the band gap energy.
For an insulator (Egap≈4eV) at room temperature (T ~ 293 K), the probability is:
so the electron better get used to being totally bound to its parent atom.
For a semi-conductor (Egap≈1eV) the odds are much better:
You may think that a one in a billion chance is pretty small odds, but remember that there are about 1022 atoms in a single gram of silicon. At these odds, quite a few electrons will win the lottery and be free. Of course, in a conductor every atom contributes a free electron so although an intrinsic semiconductor will conduct current, its conductivity is quite a bit less than a typical metal.
One interesting property of intrinsic semiconductors, in contrast to conductors, is their very strong temperature dependence. Semiconductors become exponentially more conductive with increasing temperature while conductors typically become linearly less conductive with temperature. This allows semiconductors to be very effective as thermistors. For example, over a temperature range from 0 C to 100 C, the current in a semiconductor will increase by a factor of:
This enormous change in current can be easily measured allowing a very precise temperature determination.
One final feature of intrinsic semiconductors is the concept of a hole. When an electron jumps from the valence to the conduction band (a process termed thermal generation) it leaves behind a positively charged atom. Valence electrons from other atoms can then move into the vacancy (or hole) in the first atom. This process will continue with the hole gradually moving through the solid. In fact, the motion of the hole is indistinguishable from the motion of an actual positive charge carrier. This process does not occur in conductors because in a conductor all of the atoms are missing electrons and thus all of the atoms have the same positive charge. In the graphic below, the black dots represent electrons and the white dots are holes. Notice that the electron current and the hole current are always in opposite directions.
Recombination occurs when a freely moving electron meets a freely moving hole. The energy released at recombination, when the electron “falls” into the hole, is normally absorbed by neighboring atoms, but it is possible to construct a device in which this energy is released as a photon (with energy closely matching the band gap energy). This device is a light-emitting diode, or LED. Photovoltaic cells are, in essence, this process in reverse, in which an incoming photon creates an electron and hole current.
Q1
Imagine a 5.0 g crystal of pure Ge (Egap = 0.7 eV).
a. At room temperature (293 K), determine the average number of mobile electrons.
b. At room temperature (293 K), determine the average number of mobile holes.
c. At what temperature will the number of mobile charge carriers be twice the number calculated in a and b?
d. At what temperature will a 5.0 g crystal of pure Si (Egap = 1.1 eV) have the same number of mobile charge carriers as calculated in a and b?
Q2
a. Calculate the range of energy gaps that could produce visible light on recombination.
b. Consider a mole of pure semiconducting material with the gap needed to produce violet (400 nm) light. At room temperature, now many mobile electrons and holes are available to recombine into photons? What does this imply about making LEDs out of pure (non-doped) semiconducting material?
III. Doped Semiconductors
In a conductor, the density of charge carriers is fixed by the density of the material. The amazing utility of semiconductors is because this density can be altered by carefully introducing impurities into the semiconductor. This process, termed doping, allows you to tailor many of the properties of the semiconductor.
In pure silicon, each atom makes four double-bonds with its nearest neighbors. If we replace a small number of silicon atoms with, for example, antimony atoms (or any other atom with a valence of five), one of the antimony electrons will be unbound and free to move about the solid. Thus, for each antimony atom we get one free electron. Notice that this doping does not produce a corresponding hole. This type of doping creates n(egative)-type material:
Conversely, if we replace a small number of silicon atoms with, for example, boron atoms (or any other atom with a valence of three), one of the silicon electrons will be unpaired. This creates a hole that an electron from an adjacent atom will fill. This type of doping creates p(ositive)-type material:
Material is doped by either growing a crystal from molten silicon (or germanium) containing precise amounts of doping material, or by injecting the doping material into a thin surface film of the semiconductor.
Although the contribution to the current density due to the intrinsic material (via thermal generation) is highly temperature dependent, the contribution due to doping is independent of temperature. Imagine a sample of silicon doped with phosphorus at a rate of 1 per 20,000 atoms. At 273 K, consider the ratio of free electrons from phosphorus (P) to free electrons from silicon (Si):
i(P)i(Si)≈120000e−1.12(8.617x10−5)(273)
i(P)i(Si)≈700,000
compared to at 373 K:
i(P)i(Si)≈120000e−1.12(8.617x10−5)(373)
i(P)i(Si)≈1,400
A temperature change of 100 K shifts this ratio by a factor of 500.
3. A crystal of Ge is doped at a rate of 1/30000 with boron. Find the ratio of doped holes (from B) to thermal holes (from Ge) at room temperature.
IV. The p-n Junction
Imagine a sample of n-type material brought into contact with p-type material. (Remember, although there are free holes in the p-material and free electrons in the n-material, both materials are initially electrically neutral.) Very rapidly after being brought into contact, electrons will drift across the boundary and quickly combine with holes (creating fixed negative ions) and holes will rapidly drift across and combine with electrons (creating fixed positive ions), as shown below:
This process will continue until the electric field formed by the creation of the negative and positive ions (pointing left within the depletion region above) grows large enough to prevent further drifting of electrons and holes. Once this equilibrium is reached, the overall neutral material will have a “built-in” electric field in the depletion region separating the two conducting regions. This electric field will also give rise to a “built-in” potential difference between the n- and p-sides of the material. What we’d like to do now is calculate the width of the depletion region (d), as well as the electric field and electric potential in this region.
To determine the electric field, let’s apply Gauss’ law to the Gaussian surface illustrated below:
+x
The only surface with flux is the right-hand side of the gaussian rectangle, with area A. The total charge within the gaussian rectangle is the product of the doping charge density (ρ), which is negative, and the volume of the rectangle within the depletion zone (Ax). Also note that since the region of interest is not vacuum, the permittivity is given by the product of the dielectric constant (κ) and ε0.
Putting this all together yields,
∮E⋅dA=qenclosedε
\[E(A) = \frac{(-\rho)(Ax)}{\kappa\varepsilon_0\}\]
E=−ρxκε0
The negative sign indicates the electric field points to the left. This expression is only valid from x = 0 to x = d/2, since beyond d/2 the magnitude of the enclosed charge begins to decrease. By symmetry, the electric field in the depletion region looks like this:
Now that we know the electric field in the depletion zone we can determine an expression for the potential difference across the depletion zone.
ΔV=−∈d0E⋅dx
By symmetry, we can integrate from 0 to d/2 and double the result:
ΔV=2(i∫d/20E⋅dx)
ΔV=2(−∫d/20−ρxκε0dx)
ΔV=2(ρ(d2)22κε0−0)
ΔV=ρd24κε0
Thus, the potential within the depletion zone looks like this:
The fundamental remaining question is how large will the potential difference grow before the junction reaches equilibrium. To determine this, let’s examine the p-material:
• In order for a hole to roll “uphill” into the n-material it would need to have a kinetic energy at least as large as eV. The probability of this occurring is given by the Boltzmann factor with energy eV. If this did occur, the potential difference would increase. This flow of charge is referred to as the recombination current, iR, and is given by:
iR≈Nholese−e\DeltaVkT
• On the other hand, it is also possible for a thermally generated electron to form in the p-material and freely roll uphill into the n-material (remember, negative charges roll “up” electric potentials). The probability of this occurring is given by the Boltzmann factor with energy Egap/2. If this occurred, the potential difference would decrease. This flow of charge is referred to as the thermal generation current, iTh, and is given by:
iTh≈Nholese−Egap2kT
At equilibrium, these two currents must be equal. Of course, the same processes can occur in the n-material and, assuming symmetric doping, equilibrium will occur at the same potential difference.
Finally, we can calculate some stuff! Let’s imagine a room temperature silicon p-n junction doped (on both sides) at 1 part per 40,000 atoms. The density of silicon is 2.4 g/cm3, silicon has 28 g/mol, and the dielectric constant for silicon is 12.
First, let’s calculate the internal potential difference across the junction. Setting the two currents equal leads to:
iR=iTh
\[N_{holes} e^{-\frac{e\Delta V}{kT} = N_{atoms} e^{-\frac{E_{GAP}}{2kT}}\]
\[1e^{-\frac{e\Delta V}{kT} = 40000e^{\frac{E_{gap}}{2kT}\]
e(Egap2−eΔV)/kT=40000
(Egap2−eΔV)/kT=ln(40000)
eΔV=Egap2−kTln(40000)
eΔV=0.29\texeV
ΔV=0.29 V
Since the doping charge density is:
\[\rho = \bigg( \frac{1.6\times 10^{-19)C}{40000\text{ atoms}}\bigg)\bigg( \frac{6.02\times 10^{-23}\text{ atoms}}{mol}\
the width of the depletion zone is:
The depletion zone is only about 24 nm wide at equilibrium.
The maximum magnitude electric field, occurring at the boundary between the p- and n-material, is
The electric field at the boundary is a whopping 24 megavolts per meter.
4. Imagine a room temperature germanium p-n junction doped (on both sides) at 1018 atoms per cm3. The density of germanium is 5.4 g/cm3, germanium has 73 g/mol, and the dielectric constant for germanium is 16. Calculate the internal potential difference, the width of the depletion zone, and the maximum electric field in the junction.
V. The Diode
Now that we understand what happens when a pn-junction is formed, our next (and final) task is to show how such a junction acts in a circuit. First, let’s consider a pn-junction under reverse bias conditions, in which the positive supply voltage is attached to the n-material.
In this configuration, the bias voltage, Vb, adds to the internal potential difference of the junction effectively making the potential “hill” higher:
Now consider how this affects the two currents flowing through the junction. We will again look at the p-material but the same conclusions can be drawn from the n-material.
• It is now more difficult for a hole to roll “uphill” into the n-material since it needs more kinetic energy, at least as large as e(V+Vb). Thus the recombination current becomes:
iR≈Nholese−e(\DeltaV+Vb)kT
• On the other hand, this has no effect on the thermal generation current, since thermally generated electrons freely roll uphill regardless of hill height. The thermal generation current is still given by:
\[i_{Th}\approx N_{atoms}e^{-\frac{E_{gap}}{2kT}\]
The total current is given by:
i=iR=−iTh
i=Nholese−e(ΔV+VbkT−iTh
i=(Nholese−e(ΔVkT)e−e(ΔVbkT−iTh
Notice that the term in parentheses is exactly equal to iTh since the junction was in equilibrium before the external voltage was applied. Thus:
i=iThe−e(ΔVbkT−iTh
i=iTh(e−e(ΔVbkT−1)
Thus, the current is negative (to the left through the junction) and very small, no larger than the thermal current. In fact, the current reaches the thermal current exponentially quickly and remains relatively constant for any bias voltage less than the break-down voltage of the device.
Now let’s consider a pn-junction under forward bias conditions, in which the positive supply voltage is attached to the p-material.
In this configuration, the bias voltage, Vb, subtracts from the internal potential difference of the junction effectively making the potential “hill” smaller:
Now consider how this affects the two currents flowing through the junction.
• It is now easier for a hole to roll “uphill” into the n-material since it needs less kinetic energy, now only e(V-Vb). Thus the recombination current becomes:
iR≈Nholese−e(ΔV+VbkT
• Again, this has no effect on the thermal generation current, since thermally generated electrons freely roll uphill regardless of hill height.
The total current is given by:
i=iR−iTh
i=Nholese−e(ΔV−VbkT−iTh
i=(Nholese−e(ΔVkT)e+e(ΔVbkT−iTh
i=iThe+e(ΔVbkT−iTh
i=iTh(e+e(ΔVkT−1)
Thus, the current is positive (to the right through the junction) and grows exponentially large with applied voltage.
The two results can be combined into the Shockley Diode Equation,
\[i =i_{Th}(e^{\pm\frac{eV_b}{kT}-1)\]
where the positive sign is chosen for forward and the negative sign for reverse bias.
Q5
a. Estimate the ratio of forward-bias (+0.5 V) current to reverse-bias (-0.5 V) current in a room temperature Ge pn-junction doped at 1/50000.
b. Estimate the same ratio at 50° C.


