4.7: X-Ray Diffraction
( \newcommand{\kernel}{\mathrm{null}\,}\)
By the end of this section, you will be able to:
- Describe interference and diffraction effects exhibited by X-rays in interaction with atomic-scale structures
Since X-ray photons are very energetic, they have relatively short wavelengths, on the order of 10−8 m to 10−12 m. Thus, typical X-ray photons act like rays when they encounter macroscopic objects, like teeth, and produce sharp shadows. However, since atoms are on the order of 0.1 nm in size, X-rays can be used to detect the location, shape, and size of atoms and molecules. The process is called X-ray diffraction, and it involves the interference of X-rays to produce patterns that can be analyzed for information about the structures that scattered the X-rays.
Perhaps the most famous example of X-ray diffraction is the discovery of the double-helical structure of DNA in 1953 by an international team of scientists working at England’s Cavendish Laboratory—American James Watson, Englishman Francis Crick, and New Zealand-born Maurice Wilkins. Using X-ray diffraction data produced by Rosalind Franklin, they were the first to model the double-helix structure of DNA that is so crucial to life. For this work, Watson, Crick, and Wilkins were awarded the 1962 Nobel Prize in Physiology or Medicine. (There is some debate and controversy over the issue that Rosalind Franklin was not included in the prize, although she died in 1958, before the prize was awarded.)
Figure 4.7.1 shows a diffraction pattern produced by the scattering of X-rays from a crystal. This process is known as X-ray crystallography because of the information it can yield about crystal structure, and it was the type of data Rosalind Franklin supplied to Watson and Crick for DNA. Not only do X-rays confirm the size and shape of atoms, they give information about the atomic arrangements in materials. For example, more recent research in high-temperature superconductors involves complex materials whose lattice arrangements are crucial to obtaining a superconducting material. These can be studied using X-ray crystallography.

Historically, the scattering of X-rays from crystals was used to prove that X-rays are energetic electromagnetic (EM) waves. This was suspected from the time of the discovery of X-rays in 1895, but it was not until 1912 that the German Max von Laue (1879–1960) convinced two of his colleagues to scatter X-rays from crystals. If a diffraction pattern is obtained, he reasoned, then the X-rays must be waves, and their wavelength could be determined. (The spacing of atoms in various crystals was reasonably well known at the time, based on good values for Avogadro’s number.) The experiments were convincing, and the 1914 Nobel Prize in Physics was given to von Laue for his suggestion leading to the proof that X-rays are EM waves. In 1915, the unique father-and-son team of Sir William Henry Bragg and his son Sir William Lawrence Bragg were awarded a joint Nobel Prize for inventing the X-ray spectrometer and the then-new science of X-ray analysis.
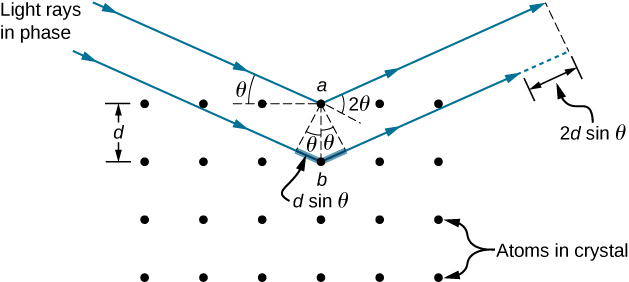
In ways reminiscent of thin-film interference, we consider two plane waves at X-ray wavelengths, each one reflecting off a different plane of atoms within a crystal’s lattice, as shown in Figure 4.7.2. From the geometry, the difference in path lengths is 2dsinθ. Constructive interference results when this distance is an integer multiple of the wavelength. This condition is captured by the Bragg equation,
mλ=2dsinθ,
for m=1,2,3,....
where m is a positive integer and d is the spacing between the planes. Following the Law of Reflection, both the incident and reflected waves are described by the same angle, θ, but unlike the general practice in geometric optics, θ is measured with respect to the surface itself, rather than the normal.
Common table salt is composed mainly of NaCl crystals. In a NaCl crystal, there is a family of planes 0.252 nm apart. If the first-order maximum is observed at an incidence angle of 18.1°, what is the wavelength of the X-ray scattering from this crystal?
Strategy:
Use the Bragg equation, Equation ???, to solve for θ.
Solution
For first-order, m=1, and the plane spacing d is known. Solving the Bragg equation for wavelength yields
λ=2dsinθm=2(0.252×10−9m)sin(18.1o)1=1.57×10−10m,or0.157nm
Significance
The determined wavelength fits within the X-ray region of the electromagnetic spectrum. Once again, the wave nature of light makes itself prominent when the wavelength (λ=0.157nm) is comparable to the size of the physical structures (d=0.252nm) it interacts with.
For the experiment described in Example 4.7.1, what are the two other angles where interference maxima may be observed? What limits the number of maxima?
- Answer
-
38.4o and 68.8o; Between θ=0o→90o, orders 1, 2, and 3, are all that exist.
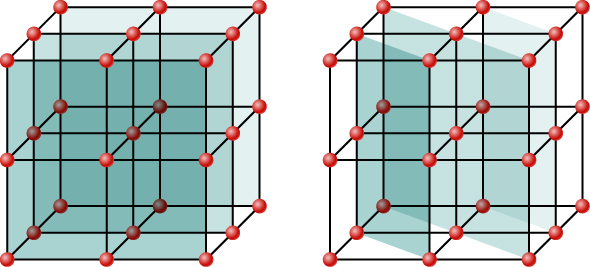
Although Figure 4.7.2 depicts a crystal as a two-dimensional array of scattering centers for simplicity, real crystals are structures in three dimensions. Scattering can occur simultaneously from different families of planes at different orientations and spacing patterns known as called Bragg planes, as shown in Figure 4.7.3. The resulting interference pattern can be quite complex.