39.6: General Questions
( \newcommand{\kernel}{\mathrm{null}\,}\)
-
Describe or sketch the different structures possible for C3H4. Do you expect these substances to have the same properties or different properties?
-
The structure of a water molecule causes it to be polar. If the water molecule had the structure illustrated below, would it still be polar? Explain.
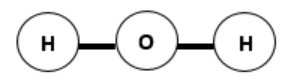
-
The flat structure of the benzene (C6H6) molecule is partly responsible for this molecule being a cancer causing substance. This molecule can slice into the DNA double helix and disrupt the cell’s ability to regulate itself. Name a substance that you think may contain the benzene molecule.

