14.6: Light Emitting Diode
( \newcommand{\kernel}{\mathrm{null}\,}\)
Let's talk about the recombining electrons for a minute. When the electron falls down from the conduction band and fills in a hole in the valence band, there is an obvious loss of energy. The question is; where does that energy go? In silicon, the answer is not very interesting. Silicon is what is known as an indirect band-gap material. What this means is that as an electron goes from the bottom of the conduction band to the top of the valence band, it must also undergo a significant change in momentum. This all comes about from the details of the band structure for the material, which we will not concern ourselves with here. As we all know, whenever something changes state, we must still conserve not only energy, but also momentum. In the case of an electron going from the conduction band to the valence band in silicon, both of these things can only be conserved if the transition also creates a quantized set of lattice vibrations, called phonons, or "heat". Phonons posses both energy and momentum, and their creation upon the recombination of an electron and hole allows for complete conservation of both energy and momentum. All of the energy which the electron gives up in going from the conduction band to the valence band (1.1 eV) ends up in phonons, which is another way of saying that the electron heats up the crystal.
In some other semiconductors, something else occurs. In a class of materials called direct band-gap semiconductors, the transition from conduction band to valence band involves essentially no change in momentum. Photons, it turns out, possess a fair amount of energy (several eV per photon in some cases) but they have very little momentum associated with them. Thus, for a direct band gap material, the excess energy of the electron-hole recombination can either be taken away as heat, or more likely, as a photon of light. This radiative transition then conserves energy and momentum by giving off light whenever an electron and hole recombine. This gives rise to (for us) a new type of device, the light emitting diode (LED). Emission of a photon in an LED is shown schematically in Figure 14.6.1.
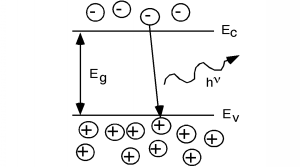 Figure 14.6.1: Radiative recombination in a direct band-gap semiconductor
Figure 14.6.1: Radiative recombination in a direct band-gap semiconductor
It was Planck who postulated that the energy of a photon was related to its frequency by a constant, which was later named after him. If the frequency of oscillation is given by the Greek letter "nu" (ν), then the energy of the photon is just hν where h is Planck's constant, which has a value of 4.14×10−15 eV⋅seconds. E=hν
When we talk about light it is conventional to specify its wavelength, λ, instead of its frequency. Visible light has a wavelength on the order of nanometers (red is about 600 nm, green about 500 nm and blue is in the 450 nm region.) A handy "rule of thumb" can be derived from the fact that λ=cν, where c is the speed of light. Since c=3×108 msec or c=3×1017 nmsec, λ(nm)=hcE(eV)=1242E(eV)
Thus, a semiconductor with a 2 eV band-gap should give off light at about 620 nm (in the range of red light). A 3 eV band-gap material would emit at 414 nm, in the violet. The human eye, of course, is not equally responsive to all colors. We show this in Figure 14.6.2, where we have also included the materials which are used for important light emitting diodes (LEDs) for each of the different spectral regions.
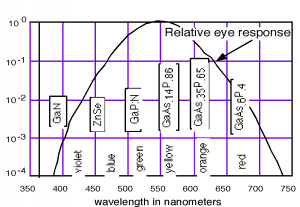 Figure 14.6.2: Relative response of the human eye to various colors
Figure 14.6.2: Relative response of the human eye to various colors
As you no doubt notice, a number of the important LEDs are based on the GaAsP system. GaAs is a direct band-gap semiconductor with a band gap of 1.42 eV (in the infrared). GaP is an indirect band-gap material with a band gap of 2.26 eV (550 nm, or green). Both As and P are group V elements. (Hence the nomenclature of the materials as III-V compound semiconductors.) We can replace some of the As with P in \mathrm{GaAs}\) and make a mixed compound semiconductor GaAs1−xPx. When the mole fraction of phosphorous is less than about 0.45 the band gap is direct, and so we can "engineer" the desired color of LED that we want by simply growing a crystal with the proper phosphorus concentration!
The properties of the GaAsP system are shown in Figure 14.6.3. It turns out that for this system, there are actually two different band gaps, as shown in the inset of Figure 14.6.3. One is a direct gap (no change in momentum) and the other is indirect. In GaAs, the direct gap has lower energy than the indirect one (like in the inset) and so the transition is a radiative one. As we start adding phosphorous to the system, both the direct and indirect band gaps increase in energy. However, the direct gap energy increases faster with phosphorous fraction than does the indirect one. At a mole fraction x of about 0.45, the gap energies cross over and the material goes from being a direct gap semiconductor to an indirect gap semiconductor. At x=0.35 the band gap is about 1.97 eV (630 nm), and so we would only expect to get light up to the red using the GaAsP system for making LEDs. Fortunately, people discovered that you could add an impurity (nitrogen) to the GaAsP system, which introduced a new level in the system. An electron could go from the indirect conduction band (for a mixture with a mole fraction greater than 0.45) to the nitrogen site, changing its momentum, but not its energy. It could then make a direct transition to the valence band, and light with colors all the way to the green became possible. The use of a nitrogen recombination center is depicted in Figure 14.6.4.
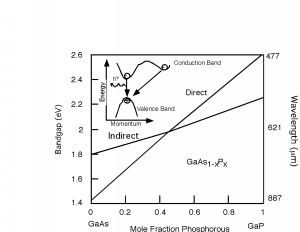 Figure 14.6.3: Band gap for the GaAsP system
Figure 14.6.3: Band gap for the GaAsP system
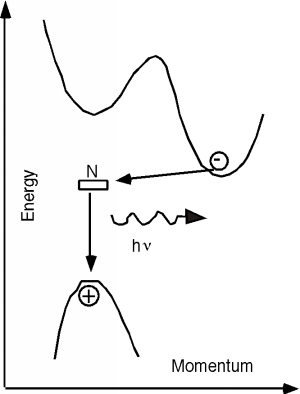 Figure 14.6.4: Addition of a nitrogen recombination center to indirect GaAsP
Figure 14.6.4: Addition of a nitrogen recombination center to indirect GaAsP
If we want colors with wavelengths shorter than the green, we must abandon the GaAsP system and look for more suitable materials. A compound semiconductor made from the II-VI elements Zn and Se make up one promising system, and several research groups have successfully made blue and blue-green LEDs from ZnSe. SiC is another (weak) blue emitter which is commercially available on the market. Recently, workers at a tiny, unknown chemical company stunned the "display world" by announcing that they had successfully fabricated a blue LED using the II-V material GaN. A good blue LED has been the "holy grail" of the display and CD-ROM research community for a number of years now. Obviously, adding blue to the already working green and red LEDs completes the set of 3 primary colors necessary for a full-color flat panel display (hang a TV screen on your wall like a picture?). Using a blue LED or laser in a CD-ROM would more than quadruple its data capacity, as bit diameter scales as λ, and hence the area as λ2.


