5.2: Thermal Expansion
( \newcommand{\kernel}{\mathrm{null}\,}\)
The Microscopic Model of Thermal Energy
Another phenomenon commonly seen that is a result of a changing temperature is the expansion/contraction of solids and liquids. While we may know that this occurs, understanding why it occurs requires a bit of review from Physics 9A.
Whenever one wants to talk about the fundamentals of thermal energy, two words immediately come up: microscopic and random. That is, we see thermal energy as the random motions of the tiny particles that comprise the macroscopic objects we see. A macroscopic indicator of the density of this random microscopic energy is what we call temperature. How does this energy manifest itself? Well, like any energy, it comes in basically two forms: potential and kinetic. We already know what the KE is, but on the microscopic level, what is the PE?
The potential energy comes from inter-particle forces that are at their core electromagnetic in nature. But there are many electric charges involved, and the potential energy function associated with this complicated combination of forces generally takes on a shape that looks like this:
Figure 5.2.1 – Molecular Potential Energy
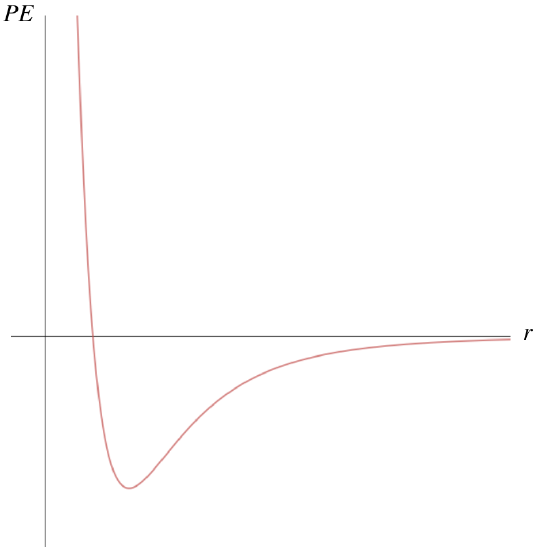
Recall that the force can be determined from the PE graph by taking the negative of the derivative (technically, the gradient):
F=−∂U∂r
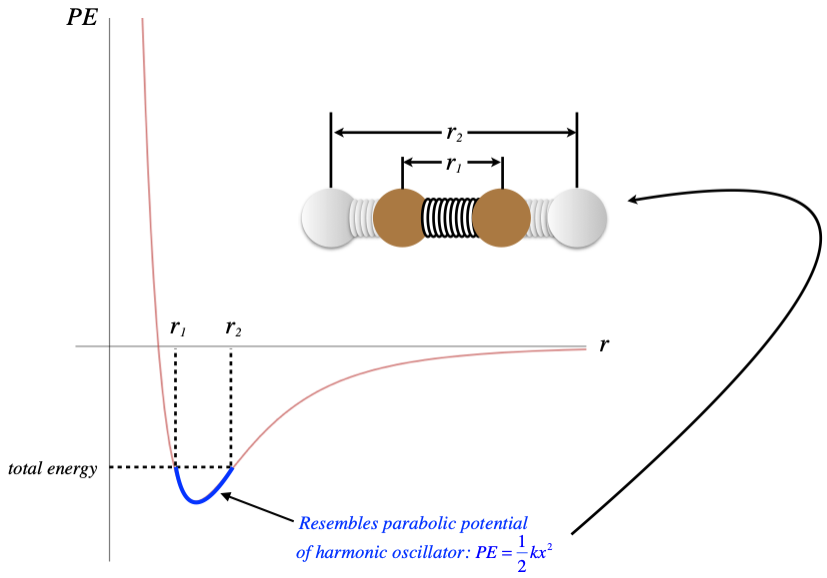
So the places on this curve that have negative slopes have positive forces, and the positive direction is in the +r-direction, which is a repulsive force. When the slope is positive, the force direction is negative, which is in the −r-direction and is therefore attractive. We therefore have a force that is alternately repulsive and attractive around a certain equilibrium position – near the bottom of the dip, usually called a potential well. The PE curve down there is closely approximated by a parabola, so the KE and PE of the particle combine to result in a physical system that looks very much like two particles bound together with a spring, with the equilibrium point of the spring being the separation of the particles associated with the bottom of the well. [Go here for a more complete review of this topic from Physics 9A.]
Figure 5.2.2 – Modeling Two-Particle Interactions with a Spring
We know that when we add energy to a spring system, the amplitude of the oscillations increases. The same is true here – the distance across the well widens. But unlike a spring system, this PE curve is not quite symmetric. When the energy of the particle-particle system is increased, the average separation actually increases.
Figure 5.2.3 – Increasing Energy Increases Average Separation
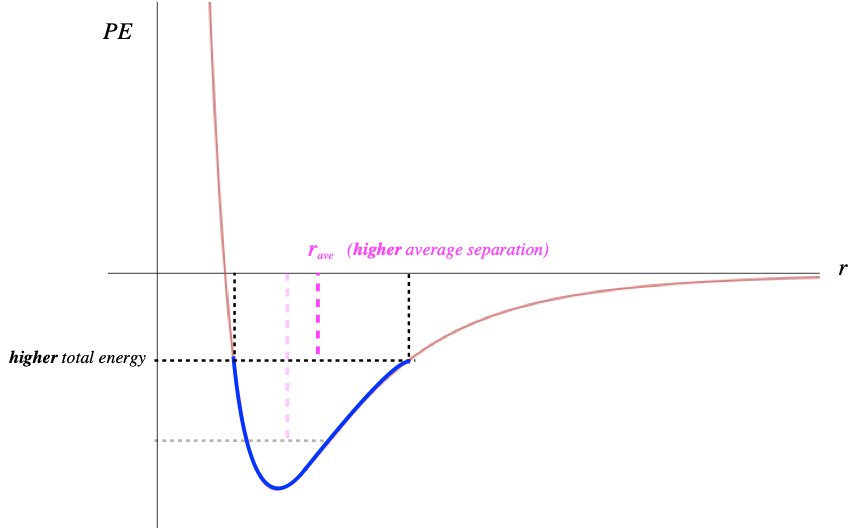
Keep in mind that when energy is added to a macroscopic system, it gets distributed through all the particles, raising all of their energies, and increasing all of their average separations. If we have N particles in a row, and the average separation increases by some small factor, then the overall length of this chain of particles increases by the same factor. While the amount of change on the microscopic level is incredibly small, it is measurable on the macroscopic level because there are so many particles.
Linear Expansion
The end result of this is that (for reasonably low temperature changes) the percentage expansion/contraction of the length of an object is proportional to the temperature change:
ΔLLo=αΔT
The constant α is called the coefficient of linear expansion, and it has units of K−1 (or, equivalently, oC−1). This is a constant that depends upon the type of material we are looking at.
Alert
We can use oC for temperature here, because we are only interested in temperature changes. The scale for kelvin and celsius are the same, only differing in their zero points. It will be a good idea as we go forward to keep close track of when these scales are interchangeable (temperature changes) and when they are not (absolute temperature related to pressure, for example).
This thermal expansion formula can be rewritten so that it relates an initial length to a final length that occurs due to a temperature change:
ΔL=αΔTLo⇒Lf−Lo=αΔTLo⇒Lf=Lo(1+αΔT)
Example 5.2.1
A structural engineer is designing the portion of a bridge where steel girders rest on top of concrete pylons, as shown in the diagram below. Space needs to be reserved between consecutive girders in the expansion joint, so that a thermal expansion of the girders will not cause them to butt against each other and buckle, and the width of the concrete pylon must be sufficient so that thermal contraction of the girder does not result in it falling off the pylon. If the distance between the centers of consecutive pylons is 60m, find the minimum width of the pylons for no buckling or slippage of the girders to occur for a temperature range between the extremes of −50oC and +50oC. The amount of thermal contraction/expansion of the concrete pylons is negligible, and the coefficient of linear expansion of steel is 1.2×10−5oC−1.

- Solution
-
The longest that a girder can be is the separation of the pylon centers, or else consecutive girders would buckle with each other. When a girder contracts due to cooling, both ends get drawn-in, so the shortest the girder can be is the pylon separation minus the pylon width, so:
Lmin=d−w⇒w=d−Lmin
The maximum expansion/contraction occurs when the temperature swings from the minimum to the maximum value (a 100oC difference), so:
ΔLLmin=αΔT⇒wd−w=αΔT
Solving for the width, we get:
w=αΔT1+αΔTd=0.00121.0012(60m)=7.2cm
Naturally in practice one would want to provide much more tolerance than this, but this is the absolute lower limit.
Volume Expansion
Of course, solids and liquids expand in more than just one dimension when their temperatures increase, and we can extend our previous analysis to determine the amount of volume expansion. For simplicity, we will consider a cube (this works with any shape, such as a sphere, but the calculus is easiest in cartesian coordinates):
Figure 5.2.4 – Infinitesimal Volume Expansion

We already know how to handle the linear expansion of each of the dimensions of this cube, so now we want to determine how much the volume expands as a result. The difference of the final and initial volumes is:
dV = V_f - V_o = \left(L_o+dL\right)^3 - L_o^3 = \left(L_o^3 + 3L_o^2dL+3L_odL^2+dL^3\right) – L_o^3 = 3L_o^2dL+3L_odL^2+dL^3
This is an infinitesimal change in the volume, so we expect an infinitesimal on the right-hand-side of the equation, but the product of multiple infinitesimals as we see in the last two terms are vanishingly small compared to the single infinitesimal, so we throw them away. We can now do a bit of algebra on what remains, to get the effect of temperature change on the volume:
\Delta V = 3L_o^2\Delta L = \dfrac{3L_o^3\Delta L}{L_o} = 3V_o\dfrac{\Delta L}{L_o} = 3V_o\left(\alpha \Delta T\right) \;\;\; \Rightarrow \;\;\; \dfrac{\Delta V}{V_o} = 3\alpha \Delta T
This looks very similar to the equation for linear expansion, with the coefficient \alpha replaced with 3\alpha. We can rename this constant "\beta" and call it the coefficient of volumetric expansion, and for all but anomalous materials (such as crystals that expand different amounts along different axes) or large temperature changes (where those other terms we threw out might become significant), it simply equals three times the coefficient of linear expansion.
Digression: The Weirdness of Water
There is one notable exception to this phenomenon, and it happens to apply to the most common substance on the Earth’s surface: water. It turns out that water near its freezing point actually expands as it cools. In addition, it expands significantly more when it changes phase to ice. If it did not, then ice would sink rather than float (which means, for example, that ponds would freeze from the bottom-up, making things significantly less comfortable for fish).
Expansion Thermometers and Thermostats
Now that we know that the expansion of a substance is proportional to its temperature change, we can get back to our idea of using temperature-based phenomena to measure temperature by using this phenomenon to fashion a thermometer. The simplest is the mercury thermometer. As the mercury changes temperature, it expands or contracts, so if we confine its expansion to be along a straight line, we can mark the container (a thin glass tube) with a linear scale and the temperature will follow that scale. Another type of thermometer involves what is called a bimetallic strip.
If we take two flat strips of different metals with different coefficients of thermal expansion and adhere them to each other, then raising their temperature will cause their lengths to expand by different amounts, resulting in the combination bending in the direction of the strip that reacts less to the temperature change (i.e. the one with a lower coefficient of linear expansion).
Figure 5.2.5 – Bimetallic Strip
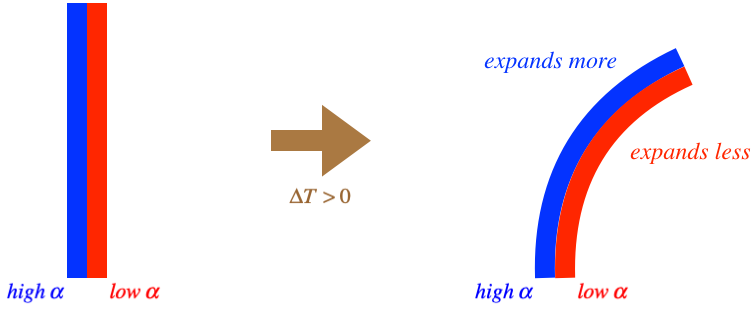
The temperature scale can therefore be measured in an angular manner – placing a scale that records the amount of bending will mirror the temperature change. An interesting application of this is the thermostat. The strip is made of a conducting material, so electric current will run through it. If we position electrical leads so that when it curls too much, the strip closes a circuit with the air conditioning, it will come on automatically. If it cools to much (curls back the other way), it can close a circuit that kicks-on a heater.


