19.15: Sigma and Pi Bonds
- Page ID
- 102034
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\) Figure \(\PageIndex{1}\) (Public Domain; the Brown Brothers, NY Times via Wikipedia)
Figure \(\PageIndex{1}\) (Public Domain; the Brown Brothers, NY Times via Wikipedia)
How many people do you think are squeezed on this street?
Our minds can handle two electrons interacting with one another in a sphere of space. But then we start putting in double bonds and triple bonds. The way we draw these bonds on paper suggests we are squeezing more electrons into the same space, and that doesn’t work. Electrons don’t like to be pushed together (especially since they all have negative charges that repel one another). So we need a more complex picture that works for all these electrons.
Sigma and Pi Bonds
The hybridization model helps explain molecules with double or triple bonds (see figure below). Ethene \(\left( \ce{C_2H_4} \right)\) contains a double covalent bond between the two carbon atoms, and single bonds between the carbon atoms and the hydrogen atoms. The entire molecule is planar.
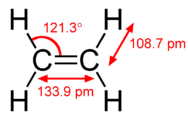 Figure \(\PageIndex{2}\): Geometry of ethene molecule. (Public Domain; Ben Mills (Wikimedia: Benjah-bmm27) via Wikipedia)
Figure \(\PageIndex{2}\): Geometry of ethene molecule. (Public Domain; Ben Mills (Wikimedia: Benjah-bmm27) via Wikipedia)
As can be seen in the figure below, the electron domain geometry around each carbon independently is trigonal planar. This corresponds to \(sp^2\) hybridization. Previously, we saw carbon undergo \(sp^3\) hybridization in a \(\ce{CH_4}\) molecule, so the electron promotion is the same for ethene, but the hybridization occurs only between the single \(s\) orbital and two of the three \(p\) orbitals. This generates a set of three \(sp^2\) hybrids, along with an unhybridized \(2p_z\) orbital. Each contains one electron and so is capable of forming a covalent bond.
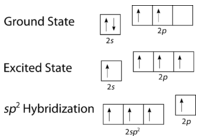 Figure \(\PageIndex{3}\): Hybridization in ethene. (CC BY-NC 3.0; Joy Sheng via CK-12 Foundation)
Figure \(\PageIndex{3}\): Hybridization in ethene. (CC BY-NC 3.0; Joy Sheng via CK-12 Foundation)
The three \(sp^2\) hybrid orbitals lie in one plane, while the unhybridized \(2p_z\) orbital is oriented perpendicular to that plane. The bonding in \(\ce{C_2H_4}\) is explained as follows: one of the three \(sp^2\) hybrids forms a bond by overlapping with the identical hybrid orbital on the other carbon atom. The remaining two hybrid orbitals form bonds by overlapping with the \(1s\) orbital of a hydrogen atom. Finally, the \(2p_z\) orbitals on each carbon atom form another bond by overlapping with one another sideways.
It is necessary to distinguish between the two types of covalent bonds in a \(\ce{C_2H_4}\) molecule. A sigma bond (\(\sigma\) bond) is a bond formed by the overlap of orbitals in an end-to-end fashion, with the electron density concentrated between the nuclei of the bonding atoms. A pi bond (\(\pi\) bond) is a bond formed by the overlap of orbitals in a side-by-side fashion with the electron density concentrated above and below the plane of the nuclei of the bonding atoms. The figure below shows the two types of bonding in \(\ce{C_2H_4}\). The \(sp^2\) hybrid orbitals are purple and the \(p_z\) orbital is blue. Three sigma bonds are formed from each carbon atom for a total of six sigma bonds in the molecule. The pi bond is the "second" bond of the double bonds between the carbon atoms, and is shown as an elongated green lobe that extends both above and below the plane of the molecule. This plane contains the six atoms and all of the sigma bonds.
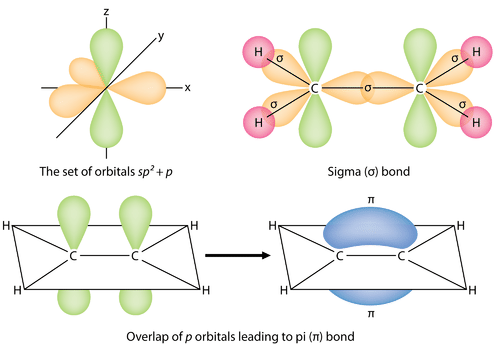 Figure \(\PageIndex{4}\): Sigma and pi bonds. (CC BY-NC 3.0; Zachary Wilson via CK-12 Foundation)
Figure \(\PageIndex{4}\): Sigma and pi bonds. (CC BY-NC 3.0; Zachary Wilson via CK-12 Foundation)
In a conventional Lewis electron-dot structure, a double bond is shown as a double dash between the atoms, as in \(\ce{C=C}\). It is important to realize, however, that the two bonds are different: one is a sigma bond, while the other is a pi bond.
Ethyne \(\left( \ce{C_2H_2} \right)\) is a linear molecule with a triple bond between the two carbon atoms (see figure below). The hybridization is therefore \(sp\).
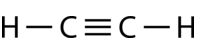 Figure \(\PageIndex{5}\): Ethyne structure. (CC BY-NC 3.0; Joy Sheng via CK-12 Foundation)
Figure \(\PageIndex{5}\): Ethyne structure. (CC BY-NC 3.0; Joy Sheng via CK-12 Foundation)
The promotion of an electron in the carbon atom occurs in the same way. However, the hybridization now involves only the \(2s\) orbital and the \(2p_x\) orbital, leaving the \(2p_y\) and the \(2p_z\) orbitals unhybridized.
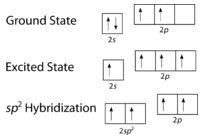 Figure \(\PageIndex{6}\): Hybridization in ethyne. (CC BY-NC 3.0; Joy Sheng via CK-12 Foundation)
Figure \(\PageIndex{6}\): Hybridization in ethyne. (CC BY-NC 3.0; Joy Sheng via CK-12 Foundation)
The \(sp\) hybrid orbitals form a sigma bond between each other as well as sigma bonds to the hydrogen atoms. Both the \(p_y\) and the \(p_z\) orbitals on each carbon atom form pi bonds between each other. As with ethene, these side-to-side overlaps are above and below the plane of the molecule. The orientation of the two pi bonds is that they are perpendicular to one another (see figure below). One pi bond is above and below the line of the molecule as shown, while the other is in front of and behind the page.
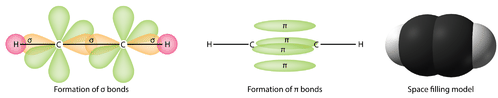 Figure \(\PageIndex{7}\): The \(\ce{C_2H_2}\) molecule contains a triple bond between the two carbon atoms, one of which is a sigma bond, and two of which are pi bonds. (CC BY-NC 3.0 (3D model available under public domain; Zachary Wilson, using 3D model by Ben Mills (User: Benjah-bmm27/Wikimedia Commons) via CK-12 Foundation; 3D model: http://commons.wikimedia.org/wiki/File:Acetylene-CRC-IR-3D-vdW.png))
Figure \(\PageIndex{7}\): The \(\ce{C_2H_2}\) molecule contains a triple bond between the two carbon atoms, one of which is a sigma bond, and two of which are pi bonds. (CC BY-NC 3.0 (3D model available under public domain; Zachary Wilson, using 3D model by Ben Mills (User: Benjah-bmm27/Wikimedia Commons) via CK-12 Foundation; 3D model: http://commons.wikimedia.org/wiki/File:Acetylene-CRC-IR-3D-vdW.png))
In general, single bonds between atoms are always sigma bonds. Double bonds are comprised of one sigma and one pi bond. Triple bonds are comprised of one sigma bond and two pi bonds.
Summary
- Sigma bonds form between two atoms.
- Pi bonds form from p orbital overlap.
Review
- What is the hybridization around each carbon in ethene?
- What are the two types of bonds in C=C?
- What is the shape of the ethene molecule?
- How are the ethyne pi bonds oriented in relation to each other?

