7.A: Quantum Mechanics (Answers)
- Page ID
- 10332
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Check Your Understanding
7.1. \(\displaystyle (3+4i)(3−4i)=9−16i^2=25\)
7.2. \(\displaystyle A=\sqrt{2/L}\)
7.3. \(\displaystyle (1/2−1/π)/2=9%\)
7.4. \(\displaystyle 4.1×10^{−8}eV; 1.1×10^{−5}nm\)
7.5. \(\displaystyle 0.5mω^2x^2ψ(x)∗ψ(x)\)
7.6. None. The first function has a discontinuity; the second function is double-valued; and the third function diverges so is not normalizable.
7.7. a. 9.1%;
b. 25%
7.8. a. 295 N/m;
b. 0.277 eV
7.9. \(\displaystyle ⟨x⟩=0\)
7.10. \(\displaystyle L_{proton}/L_{electron}=\sqrt{m_e/m_p}=2.3%\)
Conceptual Questions
1. \(\displaystyle 1/\sqrt{L}\), where \(\displaystyle L=length\); 1/L, where \(\displaystyle L=length\)
3. The wave function does not correspond directly to any measured quantity. It is a tool for predicting the values of physical quantities.
5. The average value of the physical quantity for a large number of particles with the same wave function.
7. Yes, if its position is completely unknown. Yes, if its momentum is completely unknown.
9. No. According to the uncertainty principle, if the uncertainty on the particle’s position is small, the uncertainty on its momentum is large. Similarly, if the uncertainty on the particle’s position is large, the uncertainty on its momentum is small.
11. No, it means that predictions about the particle (expressed in terms of probabilities) are time-independent.
13. No, because the probability of the particle existing in a narrow (infinitesimally small) interval at the discontinuity is undefined.
15. No. For an infinite square well, the spacing between energy levels increases with the quantum number n. The smallest energy measured corresponds to the transition from n = 2 to 1, which is three times the ground state energy. The largest energy measured corresponds to a transition from \(\displaystyle n=∞\) to 1, which is infinity. (Note: Even particles with extremely large energies remain bound to an infinite square well—they can never “escape”)
17. No. This energy corresponds to \(\displaystyle n=0.25\), but n must be an integer.
19. Because the smallest allowed value of the quantum number n for a simple harmonic oscillator is 0. No, because quantum mechanics and classical mechanics agree only in the limit of large nn.
21. Yes, within the constraints of the uncertainty principle. If the oscillating particle is localized, the momentum and therefore energy of the oscillator are distributed.
23. doubling the barrier width
25. No, the restoring force on the particle at the walls of an infinite square well is infinity.
Problems
27. \(\displaystyle ∣ψ(x)∣^2sin^2ωt\)
29. (a) and (e), can be normalized
31. a. \(\displaystyle A=\sqrt{2α/π}\);
b. \(\displaystyle probability=29.3%\);
c. \(\displaystyle ⟨x⟩=0⟨x⟩=0\);
d. \(\displaystyle ⟨p⟩=0\);
e. \(\displaystyle ⟨K⟩=α^2ℏ^2/2m\)
33. a. \(\displaystyle Δp≥2.11×10^{−34}N⋅s\);
b. \(\displaystyle Δv≥6.31×10^{−8}m\);
c. \(\displaystyle Δv/\sqrt{k_BT/m_α}=5.94×10^{−11}\)
35. \(\displaystyle Δτ≥1.6×10^{−25}s\)
37. a. \(\displaystyle Δf≥1.59MHz\);
b. \(\displaystyle Δω/ω_0=3.135×10^{−9}\)
39. Carrying out the derivatives yields \(\displaystyle k^2=\frac{ω^2}{c^2}\).
41. Carrying out the derivatives (as above) for the sine function gives a cosine on the right side the equation, so the equality fails. The same occurs for the cosine solution.
43. \(\displaystyle E=ℏ^2k^2/2m\)
45. \(\displaystyle ℏ^2k^2ℏ\); The particle has definite momentum and therefore definite momentum squared.
47. 9.4 eV, 64%
49. 0.38 nm
51. 1.82 MeV
53. 24.7 nm
55. \(\displaystyle 6.03Å\)
57. a.
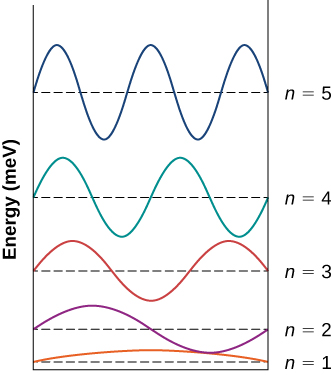
The wave functions for the n=1 through n=5 states of the electron in an infinite square well are shown. Each function is displaced vertically by its energy, measured in meV. The n=1 state is the first half wave of the sine function. The n=2 function is the first full wave of the sine function. The n=3 function is the first one and a half waves of the sine function. The n=4 function is the first two waves of the sine function. The n=5 function is the first two and a half waves of the sine function. ;
b. \(\displaystyle λ_{5→3}=12.9nm,λ_{3→1}=25.8nm,λ_{4→3}=29.4nm\)
59. proof
61. \(\displaystyle 6.662×10^{14}Hz\)
63. \(\displaystyle n≈2.037×10^{30}\)
65. \(\displaystyle ⟨x⟩=0.5mω^2⟨x^2⟩=ℏω/4\); \(\displaystyle ⟨K⟩=⟨E⟩−⟨U⟩=ℏω/4\)
67. proof
69. A complex function of the form, \(\displaystyle Ae^{iϕ}\), satisfies Schrӧdinger’s time-independent equation. The operators for kinetic and total energy are linear, so any linear combination of such wave functions is also a valid solution to Schrӧdinger’s equation. Therefore, we conclude that Equation 7.68 satisfies Equation 7.61, and Equation 7.69 satisfies Equation 7.63.
71. a. 4.21%;
b. 0.84%;
c. 0.06%
73. a. 0.13%;
b. close to 0%
75. 0.38 nm
Additional Problems
77. proof
79. a. 4.0 %;
b. 1.4 %;
c. 4.0%;
d. 1.4%
81. a. \(\displaystyle t=mL^2/h=2.15×10^{26}years\);
b. \(\displaystyle E_1=1.46×10^{−66}J,K=0.4J\)
83. proof
85. 1.2 N/m
87. 0
Challenge Problems
89. 19.2µm;11.5µm19.2µm;11.5µm
91. 3.92%
93. proof


