8.4: Electron Spin
( \newcommand{\kernel}{\mathrm{null}\,}\)
By the end of this section, you will be able to:
- Express the state of an electron in a hydrogen atom in terms of five quantum numbers
- Use quantum numbers to calculate the magnitude and direction of the spin and magnetic moment of an electron
- Explain the fine and hyperfine structure of the hydrogen spectrum in terms of magnetic interactions inside the hydrogen atom
In this section, we consider the effects of electron spin. Spin introduces two additional quantum numbers to our model of the hydrogen atom. Both were discovered by looking at the fine structure of atomic spectra. Spin is a fundamental characteristic of all particles, not just electrons, and is analogous to the intrinsic spin of extended bodies about their own axes, such as the daily rotation of Earth.
Spin is quantized in the same manner as orbital angular momentum. It has been found that the magnitude of the intrinsic spin angular momentum S of an electron is given by
S=√s(s+1)ℏ,
where s is defined to be the spin quantum number. This is similar to the quantization of L, except that the only value allowed for s for an electron is s=1/2. The electron is said to be a “spin-half particle.” The spin projection quantum number ms is associated with the z-components of spin, expressed by
Sz=msℏ.
In general, the allowed quantum numbers are
ms=−s,−s+1,...,0,...,+s−1,s.
For the special case of an electron (s=1/2),
ms=−12 or 12.
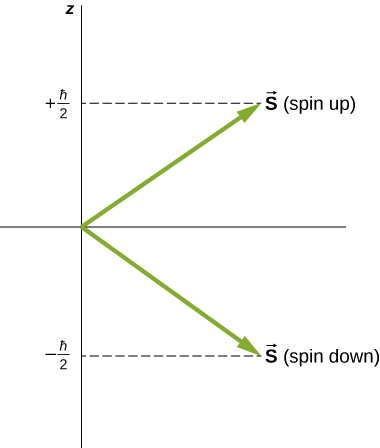
Directions of intrinsic spin are quantized, just as they were for orbital angular momentum. The ms=−1/2 state is called the “spin-down” state and has a z-component of spin, sz=−1/2, the ms=+1/2 state is called the “spin-up” state and has a z-component of spin, sz=+1/2. These states are shown in Figure 8.4.1.
The intrinsic magnetic dipole moment of an electron μe can also be expressed in terms of the spin quantum number. In analogy to the orbital angular momentum, the magnitude of the electron magnetic moment is
μs=(e2me)S.
According to the special theory of relativity, this value is low by a factor of 2. Thus, in vector form, the spin magnetic moment is
→μ=(eme)→S.
The z-component of the magnetic moment is (from Equation ???)
μz=−(eme)Sz=−(eme)msℏ.
The spin projection quantum number has just two values (ms=±1/2), so the z-component of the magnetic moment also has just two values:
μz=±(e2me)=±μB,
where μB is one Bohr magneton. An electron is magnetic, so we expect the electron to interact with other magnetic fields. We consider two special cases: the interaction of a free electron with an external (nonuniform) magnetic field, and an electron in a hydrogen atom with a magnetic field produced by the orbital angular momentum of the electron.
Electron in an External Field
The Stern-Gerlach experiment provides experimental evidence that electrons have spin angular momentum. The experiment passes a stream of silver (Ag) atoms through an external, nonuniform magnetic field. The Ag atom has an orbital angular momentum of zero and contains a single unpaired electron in the outer shell. Therefore, the total angular momentum of the Ag atom is due entirely to the spin of the outer electron (s=1/2). Due to electron spin, the Ag atoms act as tiny magnets as they pass through the magnetic field. These “magnets” have two possible orientations, which correspond to the spin-up and -down states of the electron. The magnetic field diverts the spin up atoms in one direction and the spin-down atoms in another direction. This produces two distinct bands on a screen (Figure 8.4.2).
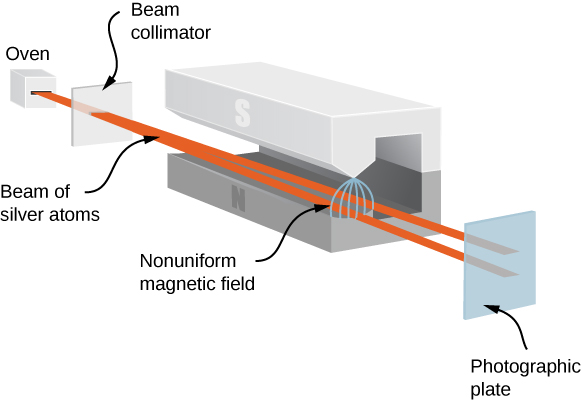
According to classical predictions, the angular momentum (and, therefore, the magnetic moment) of the Ag atom can point in any direction, so one expects, instead, a continuous smudge on the screen. The resulting two bands of the Stern-Gerlach experiment provide startling support for the ideas of quantum mechanics.
Visit PhET Explorations: Stern-Gerlach Experiment to learn more about the Stern-Gerlach experiment.
A hydrogen atom in the ground state is placed in an external uniform magnetic field (B=1.5T). Determine the frequency of radiation produced in a transition between the spin-up and spin-down states of the electron.
Strategy
The spin projection quantum number is ms=±1/2, so the z-component of the magnetic moment is
μz=±(e2me)=±μBℏ.
The potential energy associated with the interaction between the electron magnetic moment and the external magnetic field is
U=−μzB=∓μBB.
The frequency of light emitted is proportional to the energy (ΔE) difference between these two states.
Solution
The energy difference between these states is ΔE=2μBB, so the frequency of radiation produced is
f=ΔEh=2μBBh=2(5.79×10−5eVT)(1.5T)4.136×10−15eV⋅s=4.2×1010cycless.
Significance
The electron magnetic moment couples with the external magnetic field. The energy of this system is different whether the electron is aligned or not with the proton. The frequency of radiation produced by a transition between these states is proportional to the energy difference. If we double the strength of the magnetic field, holding all other things constant, the frequency of the radiation doubles and its wavelength is cut in half.
If the Stern-Gerlach experiment yielded four distinct bands instead of two, what might be concluded about the spin quantum number of the charged particle?
- Answer
-
s=3/2<
Spin-Orbit Coupling (Fine Structure)
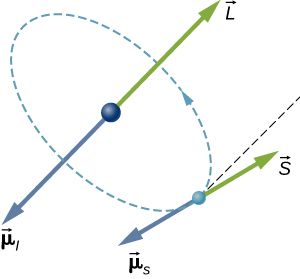
In a hydrogen atom, the electron magnetic moment can interact with the magnetic field produced by the orbital angular momentum of the electron, a phenomenon called spin-orbit coupling. The orbital angular momentum (→L), orbital magnetic moment (→μ), spin angular momentum (→S), and spin magnetic moment (→μs) vectors are shown together in Figure 8.4.3.
Just as the energy levels of a hydrogen atom can be split by an external magnetic field, so too are the energy levels of a hydrogen atom split by internal magnetic fields of the atom. If the magnetic moment of the electron and orbital magnetic moment of the electron are antiparallel, the potential energy from the magnetic interaction is relatively high, but when these moments are parallel, the potential energy is relatively small. Transition from each of these two states to a lower-energy level results in the emission of a photon of slightly different frequency. That is, the spin-orbit coupling “splits” the spectral line expected from a spin-less electron. The fine structure of the hydrogen spectrum is explained by spin-orbit coupling.
Electron Spin-Nuclear Spin Coupling (Hyperfine Structure)
Just like an electron, a proton is spin 1/2 and has a magnetic moment. (According to nuclear theory, this moment is due to the orbital motion of quarks within the proton.) The hyperfine structure of the hydrogen spectrum is explained by the interaction between the magnetic moment of the proton and the magnetic moment of the electron, an interaction known as spin-spin coupling. The energy of the electron-proton system is different depending on whether or not the moments are aligned. Transitions between these states (spin-flip transitions) result in the emission of a photon with a wavelength of λ=21 cm (in the radio range). The 21-cm line in atomic spectroscopy is a “fingerprint” of hydrogen gas. Astronomers exploit this spectral line to map the spiral arms of galaxies, which are composed mostly of hydrogen (Figure 8.4.4).

A complete specification of the state of an electron in a hydrogen atom requires five quantum numbers: n, l, m, s, and ms. The names, symbols, and allowed values of these quantum numbers are summarized in Table 8.4.4.
| Name | Symbol | Allowed values |
|---|---|---|
| Principal quantum number | n | 1, 2, 3, … |
| Angular momentum | l | 0, 1, 2, … n – 1 |
| Angular momentum projection | m | 0,±1,±2,...±l |
| Spin | s | 1/2 (electrons) |
| Spin projection | ms | −1/2,+1/2 |
Note that the intrinsic quantum numbers introduced in this section (s and ms) are valid for many particles, not just electrons. For example, quarks within an atomic nucleus are also spin-half particles. As we will see later, quantum numbers help to classify subatomic particles and enter into scientific models that attempt to explain how the universe works.


